Tumour Analyses
Research Topics
Pancreatic Cancer
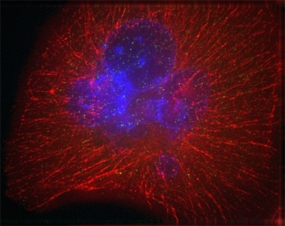
Image of a pancreatic cancer cell. DNA in the nucleus is labelled blue; red signals show the cytoskeleton
Pancreatic ductal adenocarcinoma (PDAC) is one of the most deadly cancers and most difficult to treat. It is currently the fourth and seventh most common cause of cancer-related death in the Western world (Europe and the USA) and China, respectively, although ranked only tenth in incidence, and numbers are continuously rising substantially, while they decline for most other tumour entities. Mortality is close to incidence. Only about 5% of all pancreatic cancer patients survive longer than five years and the average survival period after diagnosis is about five months. Surgical resection represents the best curative treatment approach for pancreatic cancer. However, it can only be applied to 10 to 20% of the patients.
Joining forces with leading groups in German and European pancreatic cancer research, we are aiming at establishing novel molecular diagnostic approaches for the prevention, early diagnosis, prognosis and risk stratification of pancreatic cancer. Also, we study particular functional aspects in order to improve the basic understanding of PDAC tumour biology and pathology. To this end, we look in much detail at the communication between different cell types within the tumour microenvironment, with a special focus on proteins and miRNAs. Last, we are active in identifying possible new routes for medication so as to improve on the currently dismal prognosis of the disease by means of innovative therapeutic strategies.
We pursue all this in local, national and international cooperations. Locally, we have a strong collaboration with Nathalia Giese and colleagues at the Surgery Department of Heidelberg University Clinics, which is headed by Prof. Markus Büchler. At the national level, there are several bilateral collaborations and we participated repeatedly in consortia, such as the NGFN Translational Genome Research Network Pancreatic Cancer (PaCaNet), in which major groups from Germany worked together closely on basic and clinical issues. Although PaCaNet is finished, collaboration continues. Internationally, we have been a partner in several EU-funded consortia, such as the EU COST network that covers various aspects of pancreatic cancer research.
Novel Therapy Option: DRD2 is critical for pancreatic cancer and promises pharmacological therapy by already established antagonistsr
Incidence and mortality of pancreatic ductal adenocarcinoma (PDAC) are almost equivalent, urging for the development of better therapeutic strategies. We investigated novel potential therapeutic targets for PDAC by performingglobal gene expression profiling in 195 PDAC and 41 normal pancreatic tissue samples. Superimposing the pathway context and interaction networks of aberrantly expressed genes, we identified factors with central roles in PDAC pathways. Next, tissue microarray analysis was used to verify the expression of the candidate targets in an independent set of 152 samples comprising 40 normal pancreatic tissues, 63 PDAC sections and 49 samples of chronic pancreatitis.
We identified dopamine receptor D2 (DRD2) as a key modulator of cancer pathways in PDAC. DRD2 up-regulation at the protein level was validated in a large independent sample cohort (n=152). Most importantly, we found that blockade of DRD2, through RNAi or pharmacological inhibition using FDA-approved antagonists, such as Haloperidol, hampers the proliferative and invasive capacities of pancreatic cancer cells in vitro and in vivo while modulating cAMP and endoplasmic reticulum stress pathways. Our findings demonstrate that inhibiting DRD2 represents a novel therapeutic approach for PDAC. Given that DRD2 antagonists are currently routinely used for the management of schizophrenia, a drug-repositioning strategy could facilitate the clinical use of these agents for treating pancreatic cancer.
Publications:
Jandaghi et al. (2016) Gastroenterology 151, 1218-1231. [PDF]
Early epigenetic down-regulation of microRNA-192 expression promotes pancreatic cancer progression

Two cell lines, which express miR-192 at high level (CFPAC-1) or low level (MIAPaCa-2) were transfected with constructs that suppressed or increased miR-192 expression, respectively. Xenografted into mice, strong effects on tumour growth were observed.
Pancreatic ductal adenocarcinoma (PDAC) is characterized by very early metastasis, suggesting the hypothesis that metastasis-associated changes may occur prior to actual tumor formation. We identified miR-192 as an epigenetically regulated suppressor gene with predictive value in this disease. miR-192 was downregulated by promoter methylation in both PDAC and chronic pancreatitis (CP), the latter of which is a major risk factor for development of PDAC. Functional studies in vitro and in vivo in mouse models of PDAC showed that overexpression of miR-192 was sufficient to reduce cell proliferation and invasion. Mechanistic analyses correlated changes in miR-192 promoter methylation and expression with epithelial-mesenchymal transition (EMT). Cell proliferation and invasion were linked to altered expression of the miR-192 target gene SERPINE1 that is encoding the protein plasminogen activator inhibitor-1 (PAI-1), an established regulator of these properties in PDAC cells. Notably, our data suggested that invasive capacity was altered even before neoplastic transformation occurred, as triggered by miR-192 downregulation. Overall, our results highlighted a role for miR-192 in explaining the early metastatic behavior of PDAC and suggested its relevance as a target to develop for early diagnostics and therapy.
Publications:
Botla et al. (2016) Cancer Res. 76, 4149-4159. [PDF]
COST Action Pancreatic Cancer
Large-scale international collaboration is essential to decipher relevant information in the context of omics-scale interrogations in cancer research. This is even more important for relatively rare but very fatal diseases like pancreas cancer. The COST Action Pancreatic Cancer will facilitate the collaboration of a broad range of European and international research groups on pancreatic cancer in order to: (1) integrate knowledge and experience in a multidisciplinary way 'from molecule via cell to society', (2) promote the application of uniform study tools and protocols, (3) foster their optimal use by early-stage researchers, (4) enhance the mobility and training of researchers, and (5) disseminate the results produced to society.
The Action will develop novel tools that will improve our understanding of pancreatic cancer and its prevention, diagnosis and treatment. It also aims at answering questions related to the tumours etiology, early detection, evidence-based and personalised treatment, as well as to aspects of health management. We aim at attracting young scholars across a range of disciplines, who will work in collaboration with more experienced researchers. This will enhance active European participation in the international research efforts on pancreatic cancer, with the objective of reducing disease mortality.
For more details about partners, organisation and objectives, please go to the Action webpage.
Publications:
Milne et al. (2014) Public Health Genomics 16, 305-312. [PDF]
Synthetic-lethal screens: Depletion of Rad17 sensitises pancreatic cancer cells to gemcitabine
Chemotherapy of advanced pancreatic cancer has mainly been gemcitabine-based, but with only limited effect. Recently, combination therapy that also targets checkpoint kinase 1 (CHK1) has become an attractive option. The central role of CHK1 in many DNA damage response pathways, however, may result in undesired cytotoxicity in normal cells. We were searching for other target molecules that may be more specific and thus better suited for combination therapy.
A negative selection RNAi screen was performed, targeting over 10,000 genes. Genes that were found to be synthetically lethal with gemcitabine and whose proteins are acting upstream of CHK1 were characterised in more detail. The inhibition of RAD17 potentiated gemcitabine cytotoxicity particularly, leading to forced mitotic entry of cells arrested in S-phase by gemcitabine treatment, resulting in asymmetric DNA distribution during anaphase followed by DNA fragmentation and finally cell death by mitotic catastrophe.
Our data suggest RAD17 for gemcitabine combination therapy supplementing or complementing inhibition of checkpoint kinase 1. As opposed to CHK1, RAD17 knockdown by itself does not inhibit cell proliferation and does not lead to abnormal DNA segregation, suggesting a more specific action.
Publications:
Fredebohm et al. (2013) J. Cell Sci. 126, 3380-3389. [PDF]