Stem Cells and Cancer
- Cell and Tumor Biology

Prof. Dr. Andreas Trumpp
Division Head "Stem Cells and Cancer", Managing Director of Stem Cell Institute "HI-STEM gGmbH"
Stem cells sustain life through their ability to self-renew and regenerate tissues. Our work targets Cancer Stem Cells (CSCs) - mutated stem cells driving tumor growth, recurrence, and therapy resistance. We study how these malignant cells use plasticity to adapt and evade treatment, uncovering links between stem cell programs, tumor plasticity, and neuronal influence. Thereby, we're developing next-generation personalized precision therapies to stop CSCs to drive relapse and metastases.
Image: © Ömer Copur
Image: © Ömer Copur
Our Research
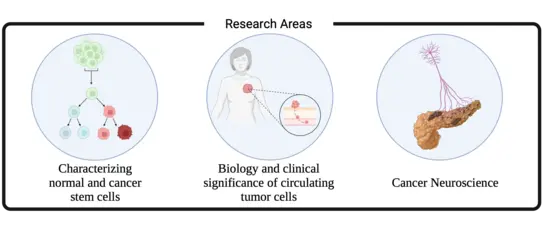
Our group investigates the biology of normal and malignant stem cells. While stem cells maintain and regenerate tissues through self-renewal and differentiation, mutations and deregulated networks transform them into cancer stem cells (CSCs). Their plasticity and clonal regenerative capacity pose major challenges in oncology including tumor growth, therapy response/resistance, relapse and metastases.
We aim to expose the molecular vulnerabilities of CSCs and stem cell mediated plasticity and translate these insights into therapeutic opportunities. Our research spans four major interconnected areas:
Hematopoietic Stem cells – mechanism of self-renewal and multipotency
Leukemic stem cells in AML – mechanisms of resistance and relapse
Circulating tumor cells and metastasis-initiating stem cells in breast cancer – understanding metastases to inform precision oncology
Cancer Neuroscience – how peripheral nerves drive pancreatic cancer progression and metastasis
We combine cutting-edge methods – including single-cell multi-omics and metabolomic technologies, multi-parameter flow cytometry and cell sorting, advanced microscopy (light-sheet), CRISPR- gene editing, engineered mouse models, patient-derived organoids and PDX models from longitudinal derived clinical samples – to bridge fundamental insights with pre-clinical and clinical translation. Our vision is to transform these discoveries into novel real time diagnostics and innovative therapy schemes that improve patient survival and quality of life.
Key Discoveries & Impact
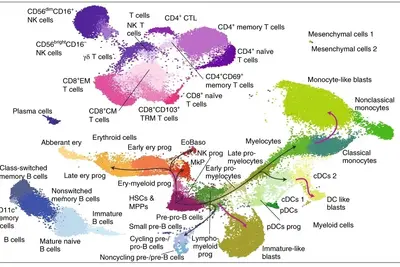
Hematopoietic stem cells (HSC) maintain lifelong blood cell production based on their capacity for extensive self-renewal and multipotency. Moreover, HSCs are critical to regenerate the blood system after stress conditions such as inflammation, blood loss or toxic insults such as chemotherapy.
→ Signature Discovery of the Trumpp team: Identified, defined and characterized “dormant” HSCs as a protected reserve, which can be activated by bone marrow stress and proposed a “Waddington-like” continuous hematopoiesis model to generate progenitors and lineage primed blood cells downstream of HSCs.
First characterization of dormant HSCs: Dormancy protects their genomic integrity and make them resistant to chemotherapy, but can be activated by inflammatory signals such as interferons (Wilson et al., Cell 2008, Essers et al., Nature 2009, Walter et al., Nature 2015)
Identified regulators of HSC dormancy and function including MYC, retinoic acid, Netrin-1, and alternative polyadenylation (Wilson et al., Genes & Development 2004, Scognamiglio et al., Cell 2016, Cabezas-Wallscheid et al., Cell 2017, Bahr et al., Nature 2018, Sommerkamp et al., Cell Stem Cell 2020).
Together with Lars Steinmetz team (EMBL), proposed the continuous “Waddington-like” hematopoiesis model – overturning the classical tree model for hematopoiesis (Velten et al., Nature Cell Biology 2017).
Revealed HSCs can function as antigen-presenting cells (Hernàndez-Malmierca et al., Cell Stem Cell 2022).
Established the MPP1-6 nomenclature for progenitors immediately downstream of HSCs (Cabezas-Wallscheid et al., Cell Stem Cell 2014, Sommerkamp et al., Blood 2021).
Identified a novel highly dormant HSC with lymphoid primed characteristics that becomes important during ageing (Lin et al., in revision 2026).
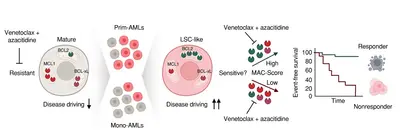
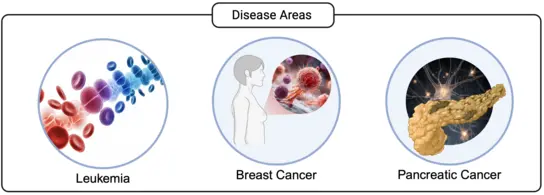
Acute myeloid leukemia (AML) is an aggressive hematologic malignancy in which disease initiation, therapy resistance, and relapse are driven by leukemic stem cells (LSCs). The LSC compartment is highly heterogeneous, a key determinant of variable therapeutic responses. This heterogeneity arises from both genetic and non-genetic factors, including somatic mutations, cell of origin, transcriptional and epigenetic states, and pronounced phenotypic plasticity. LSCs that survive therapy fuel clonal evolution, disease persistence, and relapse.
Beyond intensive and often highly toxic standard combination chemotherapies, treatment with the BCL-2 inhibitor venetoclax in combination with hypomethylating agents (e.g., 5-azacytidine) is currently transforming AML therapy and is expected to become a standard first-line treatment. We recently developed the MAC score, a personalized predictive biomarker system that enables stratification of AML patients according to their likelihood of response or resistance to venetoclax-based therapies.
→ Signature Discovery of the Trumpp team: Characterized the genetic and non-genetic heterogeneity of AML stem cells in patients at the molecular and cellular level. Developed MAC-Scoring, the first clinically useful biomarker to predict Venetoclax response in patients with high accuracy.
Identified BCAT1, controlling branched-chain amino acid catabolism, as a metabolic driver mimicking IDH/TET2 mutations, and which is upregulated in patients in response to IDH-inhibitor mediating resistance (Raffel et al., Nature 2017, Raffel et al., Blood 2020).
Together with Claudia Lengerke team (University Hospital Tübingen), uncovered LSC immune evasion and showed PARP1 inhibition restores NK surveillance (Paczulla et al., Nature 2019) – now entering clinical trials.
Tracked clonal evolution in AMLs with complex karyotype (CK-AML) with single-cell multi-omics profiling – uncovering relapse and therapy resistance mechanisms employed by LSCs (Leppä et al., Nature Genetics 2024).
Invented MAC-Scoring as a reliable predictive clinical biomarker for Venetoclax (BCL-2 inhibitor) response: a rapid (6h), affordable, and clinically applicable assay that precisely predicts individual AML patient response to VEN/HMA therapy by flow cytometric measuring expression of three BCL-2 family members in LSCs (Waclawiczek et al., Cancer Discovery 2023). MAC-Scoring is established in various Hematology-Oncology clinics in Germany, New York, and Mexico, directly guiding treatment decisions.
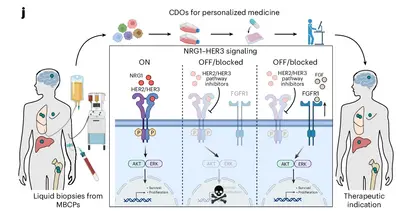
Breast cancer (BC) treatment and prognosis have been greatly improved in recent years. Nevertheless, metastatic BC (MBC) remains incurable. BC cells spread mainly to the lungs, liver, bones, lymph nodes and brain. The source of metastases is circulating tumor cells (CTCs) with metastasis-initiating stem cell potential. CTCs disseminated to the blood by the primary tumor drive metastasis, the leading cause of death in individuals with cancer including BC.
→ Signature Discovery of the Trumpp team: First to isolate metastasis stem cells directly from patient blood and established a platform to expand these cells as organoids and characterize them at the molecular and functional level associated to clinical precision oncology programs.
First experimental proof that CTCs induce metastasis and identified CD44+MET+CD47+ CTCs as drivers of bone and lung metastases (Baccelli et al., Nature Biotechnology 2013)
Created a CTC-derived organoid platform to expand CTCs from patient blood: revealing NRG1-HER2/3 and plasticity on FGFR1 as therapy escape pathways (Würth et al., Nature Cancer 2025; Schwerd-Kleine et al., Int J of Cancer 2025).
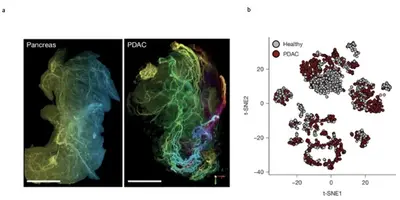
Pancreatic ductal carcinoma (PDAC) starts in the cells lining the pancreatic ducts. It is often diagnosed late and is difficult to treat, with overall 5-year survival rates around 10%. PDAC is associated to a desmoplastic stroma and hypertrophic nerves. Novel treatment schemes need to be developed to target this dismal disease.
→ Signature Discovery of the Trumpp team: Identified therapy resistance pathways and revealed the cross-talk between peripheral nerves and pancreatic cancer as a novel therapeutic target revealing new treatment concepts for PDAC.
Identified CYP3A5 as a resistance driver, now targeted with novel inhibitors (Noll et al., Nature Medicine 2016; Kuhlmann et al. Pancreas 2017).
Defined an inflammatory PDAC subtype driven by retroelement activation regulated by differential DNA methylation of the pancreatic cancer genome (Espinet et al., Cancer Discovery 2021).
The peripheral nervous system (PNS) orchestrates organ function in health and disease. Most cancers, including PDAC are infiltrated by PNS neurons. However, neuronal cell bodies reside in various PNS ganglia, far from the tumour mass. Thus, cancer-innervating or healthy-organ-innervating neurons are lacking in current tissue-sequencing datasets. To address this, the Trump team developed the Trace-n-Seq method, enabling for the first time to obtain single-cell transcriptomics data of innervating neurons of healthy pancreas and PDAC. This revealed a striking reprogramming of innervating neurons and nerves by the PDAC tumor to promote its own growth and metastasis (Thiel, Renders et al., Nature 2025).
Showed that neuronal blockade (denervation) synergizes with taxane-mediated chemo- and immunotherapy providing the basis for new clinical trials (Thiel, Renders et al., Nature 2025)
Mechanistic results obtained in the Trumpp team are directly translated into pre-clinical trials with the goal to obtain data packages for the initiation of early clinical trials. Most projects use also primary samples from patients including ones from registry studies and clinical trials. Longitudinal samples are molecularly characterized and linked to clinical data with the goal to better understand clinical response, resistance, relapse and metastasis (reverse translation).
→ Signature Discovery: Bridging lab to clinic through innovative trials in AML, breast and pancreatic cancer
Pancreatic Cancer: Led the Phase I “Intensify” trial testing CYP3A inhibitors with Paclitaxel/Gemcitabine (with Prof. Springfeld, NCT Heidelberg)
Pancreatic Cancer: Preparing Phase I/II “Synergy” Trial to combine neuronal blockade with chemo- and immunotherapy (with clinical colleagues at University Hospital Heidelberg and other German Comprehensive Cancer Centers)
Breast Cancer: Embedded in CATCH & COGNITION breast cancer registry trials (with Prof. Schneeweiss, NCT Heidelberg)
AML: Explore the effectiveness of PARP1-inhibitor and NK-cell infusion in the NAKIP-AML Phase I/II Trial (with Prof. Müller-Tidow, University Hospital Heidelberg).
AML: Prospective AML trials to further validate MAC-Scoring and generate a diagnostic Kit with diagnostic pharma and development of a Start-up company.
Future Outlook:
Cancer is not static – it is dynamic, adaptive, and deeply interconnected with its environment. By uniting stem cell (single cell-) technologies, metastasis research, and cancer neuroscience, we aim to unravel how tumors resist, recur, and spread. Our long-term vision is to develop innovative, patient-tailored strategies that target the “stemness” programs fueling cancer. From predicting drug response in AML, to pioneering neuronal blockade in pancreatic cancer, to decoding circulating tumor cells in breast cancer, our research is opening new therapeutic frontiers.
With every discovery, we move closer to transforming incurable cancers into treatable – and ultimately curable – diseases.
HI-STEM gGmbH:
In addition to his role as Head of the Division of Stem Cells and Cancer, Prof. Trumpp is also Managing Director of the Heidelberg Stem Cell Technology and Experimental Medicine (HI-STEM gGmbH), located within the main building of the DKFZ. Click here for more details.
Leadership:
Prof. Dr. Andreas Trumpp has published >200 papers in peer-reviewed journals that have been cited more than 40,000 times and he has an H-index of 93. In 2019 and 2022, he was recognized as a highly cited researcher (top 1%) by Web of Science. He received the German Cancer Award in 2020 and an ERC Advanced Grant in 2022 and is member of several scientific advisory boards and committees of scientific societies. More than 16 of his team members now run their own laboratories as group leaders or professors and many of his team members hold leading roles in industry. Prof. Trumpp’s CV can be found here.
Team
-

Prof. Dr. Andreas Trumpp
Division Head "Stem Cells and Cancer", Managing Director of Stem Cell Institute "HI-STEM gGmbH"
-

Dr. Hamed Alborzi Nia
HI-STEM Junior Research Group "Mechanisms of Cell Death and Drug Resistance"
-

Dr. Nesrine Aroua
Postdoc
-
Lucas Barea Moya
-
Tamara Benz
Postdoc
-
Moritz Berg
-

Ines Bergerweiss
PhD Student
-

Katharina Block
Trainee Technical Assistant
-
Julia Bulkescher
Technical Assistant
-
Samuele Cesaro
-

Dr. Sascha Henry Conic
Postdoc
-
Jule Dettweiler
Student Assistant
-

Dr. Elisa Donato
Postdoc
-
Daria Doncevic
-
Anna Edenhofer
PhD Student
-
Dr. Mostafa Ejtehadifar
Postdoc
-
Andrea Garcia Yuste
-

Dr. Lissy Zoe Florens Gross
Postdoc
-
Florian Grünschläger
-

Isabell Guckes
Master Student
-

Dr. Ewgenija Gutjahr
Postdoc
-
Pablo Henneman
PhD Student
-

Dr. Kim Hoa Ho
Postdoc
-

Dr. Frank Yi-Tao Huang
MD and PhD Student
-
Marlene Hühn
PhD Student
-
Nina Sophie Kruse
-
Erika Krückel
Leader Administration HI-STEM
-

Andrea Kuck
Technical Assistant
-

Dr. Christina Köhler
Clinician Scientist
-

Yara Lackousha
PhD Student
-

Sarah Lang
PhD Student
-
Aino-Maija Leppä
-
Estefania Isabella Leth
-

Jennifer Lewis
PhD Student
-

Dr. Shuiping Lin
Postdoc
-

Sabrina Lohr
Technical Assistant
-

Dr. Maria Puschhof
HI-STEM Junior Research Group "Bioinformatics"
-

Simon Renders
Clinician Scientist
-
Emma Röhm
Research Assistant
-

Sebastiaan Schieven
Postdoc
-
Dr. David Schlütermann
-

Dominique Schulz
PhD Student
-
Fabian Sieker
Master Student
-

Markus Sohn
Technical Assistant
-

Dr. Shubhankar Sood
HI-STEM Junior Research Group "Global Precision Medicine (GPM) Translational Research Group"
-

Dr. Martin Sprick
HI-STEM Research Group "Stem Cells and Metastasis"
-

Dr. Patrick Stelmach
Postdoc and Clinician Scientist
-

Natalie Stumpf
Technical Assistant
-

Karolin Stumpf
PhD Student
-

Vera Thiel
Postdoc
-
Vera Thiel
-

Dr. Marc Thier
HI-STEM Junior Research Group "Chronically progressive neurodegenerative diseases"
-
Dr. Aset Ceren Thier
-

Beatrice Tröster
PhD Student
-
William Wachter III
-

Alexander Waclawiczek
Postdoc
-

Bettina Walter
Technical Assistsant
-

Rebecca Weber
Technical Assistant
-

Dr. Jennifer Wischhusen
Postdoc
-
Dagmar Wolf
Personal Assistant
-

Nils Wörner
PhD Student
-

Dr. Roberto Würth
Postdoc and Team Leader
-
Umut Yildiz
-

Petra Zeisberger
Technical Assistant
-

Dr. Basak Özata
Business Development
Selected Publications
Thiel, V., Renders, S., et al.,
Würth, R., et al.,
Leppä, AM., et al.,
Waclawiczek, A., et al.,
Hernandez-Malmierca, P., et al.,
Espinet, E., et al.
Raffel, S. et al.
Cabezas-Wallscheid, N., et al.,
Essers, M., et al.,
Wilson, A., et al.,
Join Us
We are always looking for motivated people to join our lab.
For career programs at DKFZ check out Cancer Research Academy at DKFZ and for direct inquiries email your application to us including a cover letter stating your research interest, your CV with a complete list of your publications as well as contact details of 3 references.
Funding
Our research is generously supported by:

Get in touch with us

Prof. Dr. Andreas Trumpp
Division Head "Stem Cells and Cancer", Managing Director of Stem Cell Institute "HI-STEM gGmbH"
