Federated Information Systems
- Imaging and Radiooncology
- DKFZ Hector Cancer Institute

Prof. Dr. Martin Lablans
Head of Division
We build bridges for biomedical research with novel methods solving both technical and medico-legal data sharing issues. We apply these methods in real-world networks for joint use of data and biosamples in Europe and beyond.
Our Research
Data-driven research in the health sector depends therefore on the exploitation of distributed sources of data and tissue samples. Two significant fields of research emerge from these requirements. First, establishing the basis for carrying out research efficiently in networked federations at the technical and data content levels. Second, the implementation of regulatory requirements, which arise naturally during the processing of sensitive patient data (e.g. data protection).
In the Department of Federated Information Systems, we investigate problems that frequently arise in networked medical research:
- Semantics of data, to provide a common understanding of their meaning
- Multi-center integration of data from heterogeneous sources
- Data protection, consent management, record linkage and pseudonymization
- Distributed processes for evaluating and exchanging data
- Measurement and improvement of data quality over multiple sites
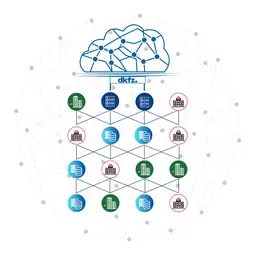
We are actively engaged in developing interoperable and reusable tools for tackling the problems in this area. In addition, we have deployed our solutions in national and international treatment federations. With our “Bridgeheads” we have created a network of exceptional partners (cf. diagram).
Projects and Networks
To build bridges for biomedical research, we pioneer novel methods solving both technical and medico-legal data sharing issues. We create real-world networks for joint use of data and biosamples in Europe and beyond.
We build bridges among institutions with GDPR-compliant federated data management solutions, including the “Mainzelliste” for pseudonymization and record linkage, several federated search solutions and the “Bridgehead” for controlled data sharing. These form the backbone of the German Cancer Consortium (DKTK), lung cancer patients data collection in the National Network Genomic Medicine (nNGM) and the “Sample Locator”, which allows federated search across more than 15 European biobanks (BBMRI-ERIC). As a bridge division to the University Medicine Mannheim, we foster data-driven collaborations as part of the DKFZ Hector Cancer Institute’s novel approach to data sharing with the Medical Informatics Initiative.
Software
Beyond concepts and prototypes, we routinely develop our methods into production-ready software tools, which we - as strong advocates of free open source software (FOSS) - publish under FOSS licences on GitHub and elsewhere.
Publications
Our Team
-

Prof. Dr. Martin Lablans
Head of Division
-
Alexandra Albu
-

Nabe Al Hasnawi
-

Moanes Ben Amor
-

Torben Brenner
-
Justin Bühler
-

Dr. David Croft
-

Pierre Delpy
-
Adrian Estevez
-
Laura Fast
-
Arturo Figueroa Macias
-

Claudia Funke
Scientific Project Coordination/Assistance
-
Tobias Gies
-
Dennis Grimm
-

Defne Halici
-
Jonathan Hirsch
-
Oleksandr Holovatyi
-
Manal Jaidar
-
Mats Johansen
-

David Juarez Guajardo-Fajardo
-
Martin Jurk
-

Jori Kern
-
Paola Klein
-

Enola Knezevic
-
Jonas Kunz
-

Dr. Tobias Kussel
-

Thomas Köhler
-

Denis Köther
-

Mohamed Lambarki
-
Marlena Meyer
-
Thewind Mom
-
Dema Salih
-
Valerie Sauer
-

Dr. Esther Schmidt
Deputy Head of Department
-
Tim Schumacher
-

Jan Skiba
-
Patrick Skowronek
-

Dr. Monique Stenzel
-

Galina Tremper
-
Govind Vijayakumar
-
Manoj Waikar
Get in touch with us

Prof. Dr. Martin Lablans
Head of Division
