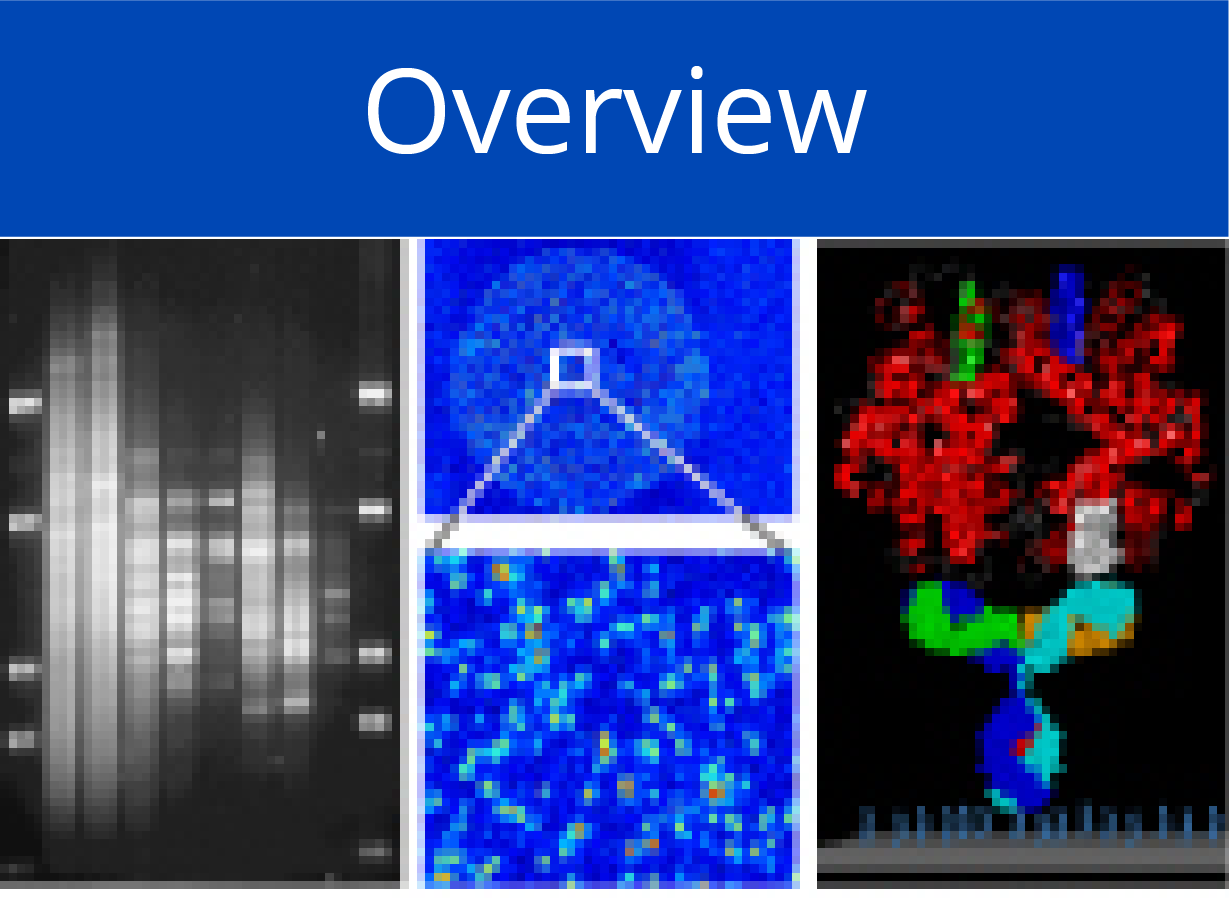
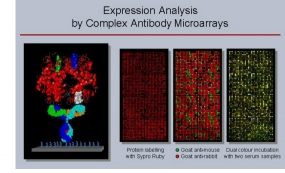
Affinity Proteomics - Antibody Microarrays
As an immediate consequence of large-scale genomic sequencing, strong interest has emerged in analysing the function of the DNA-encoded information on a similarly global scale. However, many aspects of modulation and regulation of cellular activity cannot be investigated at the level of nucleic acids but require an analysis of the proteome. Post-transcriptional control of protein translation, post-translational modifications as well as protein degradation by proteolysis, for example, have profound effects at the functional level. Estimations suggest that there are more than 200 types of protein modification. Its proportion and importance is reflected by the fact that 5% to 10% of mammalian genes encode for proteins that modify other proteins.
The complexity in the human proteome is expected to range from a hundred thousand to several million different protein molecules. With respect to data interpretation, the situation is additionally complicated by the facts that not for every protein of multicellular organisms the function is known and that there may be different functions dependent on structure variations or interacting partners. In addition, the dynamic range of protein expression is very large indeed.
Various mass spectrometry based processes exist for a powerful analysis of proteins of an organism or tissue. Also, assays such as yeast-two-hybrid analyses in all their facets permit global studies for the identification of interaction partners. Nevertheless, many other, possibly even more powerful methods are prerequisite to approaching the world of protein analysis in a manner similar to what is already possible for studies at the level of nucleic acids, and beyond.
Protein microarrays have an enormous potential of developing into a tool that will allow global characterisation of molecule mixtures at the protein level. Knowledge of genomic sequences and transcriptional profiles do not suffice for a reliable description of actual protein expression, let alone an analysis of protein structures and biochemical activities or a quantitative examination of protein-protein interactions. This kind of information, however, is crucial for a molecular understanding the biology of cells, tissues or whole organisms and has a broad biotechnical and medical potential. Utilising recently developed processes, we perfom such analyses on a large-scale with nevertheless high reproducibility, a near-single-molecule sensitivity, and an accuracy that is as good as or even superior to ELISA-based assays.
Antibody microarrays:
Utilising antibody microarrays, we pursue the analysis of studying variations in actual protein abundance, isoform occurrence and other structural variations, in particular in cells, tissues and body liquids. Basic technical processes were studied in much detail, such as appropriate surfaces, the effect of kinetics and mass transport and labelling procedures as well as many other aspects in order to establish a working system. Detailed protocols are available that allow reproducible and reliable analysis of expression variations on complex protein extracts from tissues, cells or body liquids down to attomolar concentrations. Antibody generation and selection was and is performed in collaborations with company partners as well as EU-funded initiatives that aim at the creation of well-characterised and specific antibodies or other binders (e.g., Affinomics). In addition, improvements of the preparation of protein extracts proved crucial for success. The current set-up was and is used in various projects, frequently combining the information on protein levels with other data. Also, quantification of the results is performed, either by actual counting of individual molecules or by an analysis of dissociation parameters.
Protein microarrays:
We utilise protein microarrays containing (mostly full-length) molecules for the investigation of protein-interactions in a quantitative manner. Microarray production is done by in situ synthesis by an transcription/translation process on the microarrays, starting from full-length cDNAs. Protein interaction of all kinds as well as the influence of co-factors such as small molecules can be studied this way. The largest protein array produced so far contains some 14,000 individual proteins. The current set-up was and is used in various projects for actual measurements, frequently combining the information on protein levels with other data.
Personalised proteomics:
In a recently finished technical development, we added to the in situ protein production a process that allows to present on the microarray the proteins in exactly the conformation as they occur in tissues or other samples of individual patients, reflecting all mutations or splice variants that are specific for the particular sample. Thereby, particularly the effects of individual variations on protein interaction - with other proteins, nucleic acids or samll compounds - can be studied in a quantitative manner.