Personalized Early Detection of Prostate Cancer

Prof. Dr. Peter Albers
Development of new risk-adapted strategies for the personalised early detection of prostate cancer using a baseline PSA value

Our Research
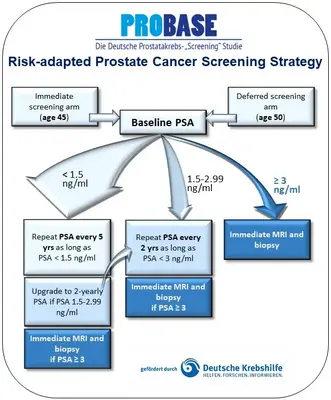
The department Personalized Early Detection of Prostate Cancer is working on new risk-adapted strategies for the personalised early detection of prostate cancer using a baseline PSA value at the age of 45 or 50. It cooperates closely with the Department of Cancer Epidemiology (Prof Rudolf Kaaks) and coordinates the PROBASE study, one of the world's largest active prostate cancer screening studies in cooperation with the DKFZ. PROBASE is a collaborative screening study with four clinical study centres at the University Hospitals of Düsseldorf, Hanover, Heidelberg and the Technical University of Munich. Data management has been coordinated at the DKFZ since the start of the study in 2013. Prof. Nikolaus Becker, who co-initiated the PROBASE study together with Prof. Peter Albers, was responsible for data management and epidemiological implementation until his retirement. The study is funded by German Cancer Aid. Since the 4th funding period (2022), the study coordination has been transferred from the University Hospital Düsseldorf to the DKFZ. The study includes reference institutions for radiology (University of Düsseldorf), nuclear medicine (University of Düsseldorf) and pathology (University of Bonn). Biobanking of blood and urine has been established at all sites. In addition, samples from radical prostatectomies are stored in a tissue biobank at the University of Bonn. An MRI image database has been set up at the University of Düsseldorf. In total, more than 50 scientists and documentation staff are currently working for PROBASE.
Prostate cancer screening is on the EU agenda to further develop risk-adapted screening programmes. PROBASE will play an important role in providing data for this approach. At national level, PROBASE data and strategies will be used to develop personalised approaches for a risk-adapted prostate cancer screening programme for Germany. The department is also involved in European initiatives on implementation strategies for prostate cancer screening programmes.
Projects

In the PROBASE study (Risk-adapted prostate cancer early detection study based on a ‘baseline’ PSA value in young men - a prospective multicenter randomised trial), a modern concept for PSA screening is being investigated. In this risk-adapted strategy, PSA tests are carried out depending on the individual risk of the man, which is determined using a baseline PSA value at the age of 45 or 50.

The aim of the European initiative with 98 participating institutions from 29 countries is to implement cancer screening programs and ensure their quality. The DKFZ is participating in a pilot study on prostate cancer screening together with the Leibniz Institute in Bremen (BIPS).
DKFZ project lead: Prof. Dr. Peter Albers
Overall project coordinator: University of Latvia (screening(at)lu.lv)
Project number: 101162959
Total funding amount: 38.7 million euros (DKFZ funding amount: 121,866 euros (incl. 26,810 euros own contribution)
Funding period: 06/2024-05/2028
Link to the European project website EUCanScreen
Participating institutions of the EUCanScreen project:
Competent Authorities:
- LATVIJAS UNIVERSITATE (LU)
- GESUNDHEIT OSTERREICH GMBH (AT)
- SCIENSANO (BE)
- HRVATSKI ZAVOD ZA JAVNO ZDRAVSTVO (HR)
- Ministry of Health of the Republic of Cyprus (CY)
- USTAV ZDRAVOTNICKYCH INFORMACI A STATISTIKY CESKE REPUBLIKY (CZ)
- REGION SYDDANMARK (DK)
- TERVISE ARENGU INSTITUUT (EE)
- TERVEYDEN JA HYVINVOINNIN LAITOS (FI)
- INSTITUT NATIONAL DU CANCER GIP (FR)
- ROBERT KOCH-INSTITUT (DE)
- MINISTRY OF HEALTH (EL)
- ORSZAGOS KORANYI PULMONOLOGIAI INTEZET (HU)
- HEILSUGAESLA HOFUDBORGARSVAEDISINS (IS)
- DEPARTMENT OF HEALTH (IE)
- ISTITUTO PER LO STUDIO E LA PREVENZIONE E LA RETE ONCOLOGICA (IT)
- LIETUVOS RESPUBLIKOS SVEIKATOS APSAUGOS MINISTERIJA (LT)
- Ministry for Health and Active Ageing - Government of Malta (MT)
- INSTITUTIA MEDICO-SANITARA PUBLICA INSTITUTUL ONCOLOGIC (MD)
- RIJKSINSTITUUT VOOR VOLKSGEZONDHEID EN MILIEU (NL)
- HELSEDIREKTORATET (NO)
- NARODOWY INSTYTUT ONKOLOGII IM. MARII SKLODOWSKIEJ-CURIE - PANSTWOWY INSTYTUT BADAWCZY (PL)
- MINISTERIO DA SAUDE - REPUBLICA PORTUGUESA (PT)
- INSTITUTUL NATIONAL DE SANATATE PUBLICA (RO)
- MINISTERSTVO ZDRAVOTNICTVA SLOVENSKEJ REPUBLIKY (SK)
- ONKOLOSKI INSTITUT LJUBLJANA (SI)
- INSTITUT CATALA D'ONCOLOGIA (ES)
- SOCIALSTYRELSEN (SE)
- STATE INSTITUTION PUBLIC HEALTH CENTER OF THE MINISTRY OF HEALTH OF UKRAINE (UA)
Affiliated Entities
- SLIMIBU PROFILAKSES UN KONTROLES CENTRS (LV)
- RIGAS STRADINA UNIVERSITATE (LV)
- PAULA STRADINA KLINISKA UNIVERSITATES SLIMNICA (LV)
- UNIVERSITAT FUR WEITERBILDUNG KREMS (AT)
- UMIT TIROL - PRIVATE UNIVERSITAT FUR GESUNDHEITSWISSENSCHAFTEN UND TECHNOLOGIE GMBH (AT)
- CENTRUM VOOR KANKEROPSPORING (BE)
- STICHTING KANKERREGISTER - FONDATION REGISTRE DU CANCER (BE)
- CENTRE COMMUNAUTAIRE DE REFERENCE POUR LE DEPISTAGE DES CANCERS (BE)
- UNIVERSITAIR ZIEKENHUIS ANTWERPEN (BE)
- KLINICKI BOLNICKI CENTAR ZAGREB (HR)
- UNIVERSITY OF CYPRUS (CY)
- TARTU ULIKOOL (EE)
- SUOMEN SYOPAYHDISTYS - CANCERFORENINGEN I FINLAND RY - CANCER SOCIETY OF FINLAND CSF (FI)
- FEDERATION HOSPITALIERE DE FRANCE (FR)
- HOSPICES CIVILS DE LYON (FR)
- UNICANCER (FR)
- ASSISTANCE PUBLIQUE HOPITAUX DE PARIS (FR)
- PLATEFORME DES DONNEES DE SANTE (FR)
- INSTITUT GUSTAVE ROUSSY (FR)
- DEUTSCHES KREBSFORSCHUNGSZENTRUM HEIDELBERG (DE)
- LEIBNIZ-INSTITUT FUR PRAVENTIONSFORSCHUNG UND EPIDEMIOLOGIE - BIPS GMBH (DE)
- DEUTSCHE KREBSGESELLSCHAFT EV (DE)
- DIOIKISI 6IS YGEIONOMIKIS PERIFEREIAS PELOPONNISOU IONION NYSON IPEIROU KAI DYTIKIS ELLADAS (EL)
- NEMZETI NEPEGESZSEGUGYI ES GYOGYSZERESZETI KOZPONT (HU)
- LANDLAEKNIS EMBAETTID (IS)
- HEALTH SERVICE EXECUTIVE (IE)
- ROYAL COLLEGE OF SURGEONS IN IRELAND (IE)
- ISTITUTO SUPERIORE DI SANITA (IT)
- MINISTERO DELLA SALUTE (IT)
- AZIENDA UNITA SANITARIA LOCALE DI REGGIO EMILIA (IT)
- REGIONE MARCHE (IT)
- AZIENDA OSPEDALIERA CITTA DELLA SALUTE E DELLA SCIENZA DI TORINO (IT)
- AZIENDA ULSS 4 VENETO ORIENTALE (IT)
- ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI (IT)
- UNIVERSITA CATTOLICA DEL SACRO CUORE (IT)
- PROVINCIA AUTONOMA DI TRENTO (IT)
- FONDAZIONE IRCCS ISTITUTO NAZIONALE DEI TUMORI (IT)
- REGIONE LOMBARDIA (IT)
- ATS PAVIA (IT)
- VIESOJI ISTAIGA VILNIAUS UNIVERSITETO LIGONINE SANTAROS KLINIKOS (LT)
- LIETUVOS SVEITAKOS MOKSLU UNIVERSITETO LIGONINE KAUNO KLINIKOS (LT)
- ERASMUS UNIVERSITAIR MEDISCH CENTRUM ROTTERDAM (NL)
- STICHTING RADBOUD UNIVERSITAIR MEDISCH CENTRUM (NL)
- STICHTING BEVOLKINGSONDERZOEK NEDERLAND (NL)
- FOLKEHELSEINSTITUTTET (NO)
- INSTYTUT GRUZLICY I CHOROB PLUC (PL)
- INSTITUTO PORTUGUES DE ONCOLOGIA DO PORTO FRANCISCO GENTIL, EPE (PT)
- UNIDADE LOCAL DE SAUDE DE SAO JOAO, E.P.E. (PT)
- UNIDADE LOCAL DE SAÚDE SANTA MARIA (PT)
- INSTITUTUL CLINIC FUNDENI (RO)
- INSTITUTUL ONCOLOGIC PROF DR ION CHIRICUTA CLUJ-NAPOCA (RO)
- NARODNY ONKOLOGICKY USTAV (SK)
- NACIONALNI INSTITUT ZA JAVNO ZDRAVJE (SI)
- ASOCIACIÓN INSTITUTO DE INVESTIGACIÓN EN SISTEMAS DE SALUD-BIOSISTEMAK (ES)
- FUNDACION PARA EL FOMENTO DE LA INVESTIGACION SANITARIA Y BIOMEDICA DE LA COMUNITAT VALENCIANA (ES)
- INSTITUTO ARAGONES DE CIENCIAS DE LA SALUD (ES)
- FUNDACION INSTITUTO DE INVESTIGACION MARQUES DE VALDECILLA (ES)
- FUNDACIO INSTITUT HOSPITAL DEL MAR D INVESTIGACIONS MEDIQUES (ES)
- SVERIGES KOMMUNER OCH REGIONER (SE)
- STATE INSTITUTION VOZIANOV INSTITUTE OF UROLOGY OF THE NATIONAL ACADEMY OF MEDICAL SCIENCES OF UKRAINE (UA)
- NATIONAL CANCER INSTITUTE (UA)
Associated Partners
- INSTITUT JULES BORDET ASBL (BE)
- MINISTERIE VAN VOLKSGEZONDHEID, WELZIJN EN SPORT (NL)
- INSTITUTO TUMORI "GIOVANNI PAOLO II" IRCCS (IT)
- AZIENDA SANITARIA LOCALE TARANTO (IT)
- MSB MEDICAL SCHOOL BERLIN GMBH (DE)
- CENTRUL NATIONAL DE SANATATE MINTALA SI LUPTA ANTODROG (RO)
- INSTITUTUL REGIONAL DE ONCOLOGIE IASI (RO)
- CENTRE BRUXELLOIS DE COORDINATION POUR LE DEPISTAGE ET LA PREVENTION DES CANCERS (BE)
Team
-

Prof. Dr. Peter Albers
-
Dr. Agne Krilaviciute
-
Dr. Petra Seibold
-
Dr. Rouvier Al-Monajjed
-
Maxime de Vrieze
Further employees
Prof. Dr. Nikolaus Becker (emeritised)
Selected Publications
Krilaviciute, A.; Kaaks, R.; Seibold, P.; De Vrieze, M.; Lakes, J.; Radtke, J. P.; Kuczyk, M.; Harke, N. N.; Debus, J.; Fink, C. A.; Herkommer, K.; Gschwend, J. E.; Meissner, V. H.; Benner, A.; Kristiansen, G.; Hadaschik, B.; Arsov, C.; Schimmöller, L.; Antoch, G.; Giesel, F. L.; Makowski, M.; Wacker, F.; Schlemmer, H. P.; Becker, N.; Albers, P.
Boschheidgen, M.; Albers, P.; Schlemmer, H. P.; Hellms, S.; Bonekamp, D.; Sauter, A.; Hadaschik, B.; Krilaviciute, A.; Radtke, J. P.; Seibold, P.; Lakes, J.; Arsov, C.; Gschwend, J. E.; Herkommer, K.; Makowski, M.; Kuczyk, M.; Wacker, F.; Harke, N. N.; Debus, J.; Koerber, S. A.; Benner, A.; Kristiansen, G.; Giesel, F. L.; Antoch, G.; Kaaks, R.; Becker, N.; Schimmöller, L.
Krilaviciute, A.; Becker, N.; Lakes, J.; Radtke, J. P.; Kuczyk, M.; Peters, I.; Harke, N. N.; Debus, J.; Koerber, S. A.; Herkommer, K.; Gschwend, J. E.; Meissner, V. H.; Benner, A.; Seibold, P.; Kristiansen, G.; Hadaschik, B.; Arsov, C.; Schimmöller, L.; Giesel, F. L.; Antoch, G.; Makowski, M.; Wacker, F.; Schlemmer, H. P.; Kaaks, R.; Albers, P.
Krilaviciute, A.; Albers, P.; Lakes, J.; Radtke, J. P.; Herkommer, K.; Gschwend, J. E.; Peters, I.; Kuczyk, M.; Koerber, S. A.; Debus, J.; Kristiansen, G.; Schimmöller, L.; Antoch, G.; Makowski, M.; Wacker, F.; Schlemmer, H. P.; Benner, A.; Giesel, F. L.; Siener, R.; Arsov, C.; Hadaschik, B.; Becker, N.; Kaaks, R.
Get in touch with us


