Cancer Drug Development
- Cell and Tumor Biology

Dr. Aubry Miller
Group Leader

Dr. Peter Nikolas Gunkel
Group Leader
The translation of new cancer research findings into novel therapeutics is fundamental to the path toward A Life Without Cancer. The Cancer Drug Development Group takes the first step on this path: discovering and developing small molecules precisely tailored to inhibit specific oncogenic proteins and target individual hallmarks of cancer.
Our Research
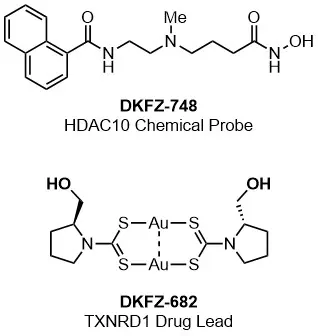
We use and share these bespoke small molecule probes as research tools to address unanswered biological questions (e.g. DKFZ-748). Additionally, we advance our inhibitors to lead substances for subsequent drug development projects (e.g. DKFZ-682). Research in our group spans target identification and validation, assay design for screening campaigns, chemical synthesis for hit-to-lead and probe optimization, and the establishment of cellular assays to evaluate the efficacy and mechanisms of action of our inhibitors.
The ideal outcome for our projects is a proof of concept in animal models, forming the foundation for development initiatives supported by public and private funding. With extensive expertise in the strategic and technical aspects of drug discovery, our team is target-class agnostic and we regularly collaborate with DKFZ colleagues seeking to translate their research into small molecule inhibitors or therapeutics.
Projects
An important reason for the limited success of genuine redox-targeting drugs in the clinic is the lack of a biomarker concept that helps to identify the cancer patients most likely to respond to ROS-inducing drugs. Using proteomics, transcriptomics, and drug sensitivity data from a large panel of non-small cell adenocarcinoma cell lines we identified a set of 15 biomarkers which accurately predict drug efficacy, both of our TXNRD1 inhibitors and related compounds, e.g. ferroptosis inducers [Samarin et al. 2023]. The gene set, which we call anti-oxidant capacity biomarkers (ACBs) and which constitutes major pathways of redox regulation and defense against oxidative stress, is tightly repressed in sensitive cells. Contrary to expectation, constitutively low ACB expression is not associated with an increased steady state level of reactive oxygen species (ROS) but a high level of nitric oxide, which is required to sustain high replication rates. ACBs are favorably expressed only in a small subset of patients in most cancer entities, with the highest percentage of patients found in leukemias and SCLC. ACBs are expressed at high levels in resistant cells due to mutations in the negative regulator of NRF2, KEAP1. Surprisingly, pharmacological inhibition of KEAP1, resulted neither in increased resistance of cancer cells to chemotherapeutics, nor to ferroptosis- or redox targeting drugs. In contrast, cells derived from normal tissues respond to pharmacological NRF2 induction by upregulating ROS scavenging enzymes and resist drug-induced apoptosis longer. This observation is fully reflected in mouse models for therapy-mediated organ damage, which show that pharmacological induction of NRF2 mitigates dose-limiting side effects. We are currently investigating whether differential regulation of ACBs in cancerous versus healthy tissue can be utilized to increase the therapeutic window of redox targeting drugs.
Histone deacetylases are a group of hydrolase enzymes which are responsible for the removal of acyl groups primarily from lysine residues on proteins, but also from other acylated amine metabolites. HDAC10, for example, is known to be a poor lysine deacetylase but an efficient polyamine (e.g. spermidine) deacetylase. Furthermore, a number of studies have highlighted HDAC10 as a potential cancer drug target: (1) High HDAC10 expression levels were found to correlate with poor clinical outcome for advanced stage 4 neuroblastoma patients who received chemotherapy. (2) Consistent with these findings, HDAC10 depletion in neuroblastoma cells interrupts autophagic flux and sensitizes cells for chemotherapy, and enforced HDAC10 expression protects neuroblastoma cells against doxorubicin treatment.
We have published the first systematic investigations into the development of HDAC10 inhibitors and have found that a number of putatively selective HDAC6 inhibitors are also very good HDAC10 inhibitors. Indeed, we found that tubastatin A, one of the best-known HDAC6 inhibitors, is an even better HDAC10 binder. More recently, we published the first fully characterized highly selective HDAC10 inhibitor chemical probes. Currently, our work is focused on the synthesis of HDAC10 PROTACs and the use of these compounds to better understand HDAC10 biology and its potential as a therapeutic target in cancer.
Kallikrein-related peptidases (KLKs) are a family of 15 secreted serine proteases that have been shown to play a role in a variety of pathological conditions. Largely investigated as biomarkers, it is now becoming increasingly clear that KLKs also have a direct role in disease progression. KLK6, for example, has been shown to promote migration and invasion of tumor cells in melanoma and colon cancers, but few reports of KLK6 inhibitors have been made in the literature.
We have recently described a novel series of depsipeptides which are covalent inhibitors of KLK6. After a thorough investigation of the SAR and MOA of these substances, we converted them into activity probes by the introduction of an alkyne handle. These probes could then be used to pull-down endogenous KLK6 from cell lines and enable a first estimation of enzymatic activity in biological samples.
Current work aims to improve these inhibitors/probes and develop a reliable diagnostic method to measure KLK6 activity in human samples.
We have accumulated extensive expertise in the synthesis and characterization of bifunctional degraders (e.g. PROTACs) over the past decade. Currently we are working on four different degrader projects, including HDAC10 PROTACs, but these projects have not yet been made public.
Team
Our team is an interdisciplinary group of cell biologists, biochemists, and synthetic organic chemists, working together to discover and develop new small molecule probes and drug leads.
- Show profile

Dr. Aubry Miller
Group Leader
-

Dr. Peter Nikolas Gunkel
Group Leader
-

Nicole de Vries
Technician
-

Franziska Deis
Technician
-
Dr. Roman Kurilov
Bioinformatician
-

Dr. Hana Nuskova
Postdoc
-

Luka Obradovic
PhD Student
-
Hannelore Pink
Technician
- Show profile

Dr. Heiko Rebmann
Postdoc
- Show profile

Nick Richert
PhD Student
-

Jasmin Rogalski
Technician
-

Dr. Sara Zahedi
Postdoc
Selected Publications
T. Marker, R.R. Steimbach, C. Perez-Borrajero, M. Luzarowski, E. Hartmann, S. Schliech, D. Pastor-Flores, E. Espinet, A. Trumpp, A.A. Teleman, F. Gräter, B. Simon, A.K. Miller*, T.P. Dick* (* = co-corresponding authors)
J. Samarin, H. Nůsková, P. Fabrowski, M. Malz, E. Amtmann, M.J. Taeubert, D. Pastor-Flores, D. Kazdal, R. Kurilov, N. de Vries, H. Pink, F. Deis, J. Hummel-Eisenbeiss, K. Kaushal, T.P. Dick, G. Hamilton, M. Muckenthaler, M. Mall, B. Lim, T. Kanamaru, G. Klinke, M. Sos, J. Frede, A.K. Miller, H. Alborzinia, N. Gunkel
R.R. Steimbach, C.J. Herbst-Gervasoni, S. Lechner, T.M. Stewart, G. Klinke, J. Ridinger, M.N.E. Geraldy, G. Tihanyi, J.R. Foley, U. Uhrig, B. Küster, G. Poschet, R.A. Casero Jr., G. Medard, I. Oehme, D.W. Christianson, N. Gunkel, A.K. Miller
S. Jänner*, D. Isak*, Y. Li, K.N. Houk, A.K. Miller
M. Morgen, P. Fabrowki, E. Amtmann, N. Gunkel, A.K. Miller
Synthesis Club
Our Synthesis Club meets every two weeks to discuss strategies and mechanisms by studying recent or classic syntheses. Feel free to contact Aubry (see below) if you would like to participate.
You can find the quizzes here.
Get in touch with us


