Computational Genomics and Systems Genetics

Prof. Dr. Oliver Stegle
Division Head
Our research group develops and applies computational approaches to study molecular variations and their phenotypic consequences.

Our Research
How does our genetic background shape phenotypic traits or cause cancer? How are genetic and environmental factors integrated at different molecular layers, and how variable are molecular states between individual cells?
We use statistical inference and machine learning as our main tools to address these scientific questions. Growing sample sizes and technological advances demand novel analytical strategies and tools that scale to datasets with millions of observations and account for spatial and temporal dependencies. We develop foundational methods in statistics and machine learning, including efficient parameter inference in models to probe genetic associations and methods for dimensionality reduction.
Find out more about our lab at https://steglelab.org/
Research Themes
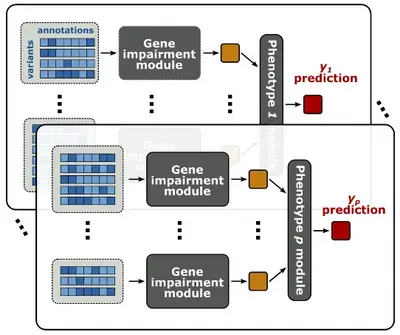
We develop statistical methods to dissect often overlooked dimensions of the genotype-phenotype relationship, including genetic effects of rare variants and indirect genetic effects.
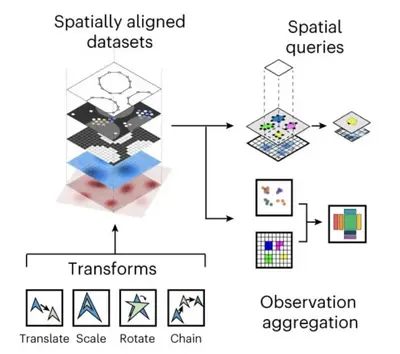
We develop foundational methods to integrate high-dimensional molecular profiles assayed in bulk or at the single-cell level. This includes innovations to allow for integrating multi-omics readouts across time and space, as well as methods for spatial omics technologies.
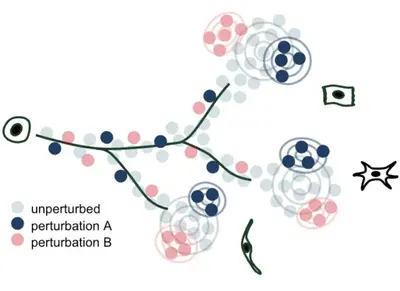
We develop methods to model genetic perturbation effects and infer causality. We leverage opportunities provided by engineered perturbations using CRISPR screens combined with single-cell readouts.
Research Groups
The MAIGE group, led by Brian Clarke, is a computational genomics group particularly interested in the application of AI and ML in a way that respects biological mechanisms and is both mathematically and statistically sound. We work with large-scale cohort data (population genetics, electronic health records) and cellular assays (single-cell omics and CRISPR perturbation assays).
MAIGE GroupTeam
We are an interdisciplinary and international team jointly hosted at the German Cancer Research Center (DKFZ) and the European Molecular Biology Laboratory (EMBL) in Heidelberg. In our team we promote openness and collaboration, and value kindness in everything we do. We take pride in every single member and their achievements and are eager to foster their scientific development.
Open Positions
We are always looking for new talent to strengthen our team!
PhD Applications:
To apply for a PhD position in our group, please refer to the DKFZ International PhD Programme.
Postdoctoral Applications:
Prospective postdoctoral researchers are encouraged to apply through the DKFZ Postdoctoral Fellowship Programme. Additionally, feel free to send your CV, a letter of motivation and ideally 2 references in a single pdf directly to Oliver Stegle.
BSc / MSc thesis or Internships:
Students interested in a BSc or MSc thesis project, or a research internship, are welcome to contact us. Please send a CV and a motivation letter tailored to the research interests of the lab, and include information about your envisioned starting date and duration.

Selected Publications
Aivazidis, A., Memi, F., Kleshchevnikov, V., Er, S., Clarke, B., Stegle, O., & Bayraktar, O. A.
Clarke, B., Holtkamp, E., Öztürk, H., Mück, M., Wahlberg, M., Meyer, K., Munzlinger, F., Brechtmann, F., Hölzlwimmer, F. R., Lindner, J., Chen, Z., Gagneur, J., & Stegle, O.
Marconato, L., Palla, G. , Yamauchi, K. A. , Virshup, I., Heidari, E., Treis, T., Toth, M., Shrestha, R. B., Vöhringer, H., Huber, W., Gerstung, M., Moore, J., Theis, F. J. , & Stegle, O.
Kleshchevnikov, V., Shmatko, A., Dann, E., Aivazidis, A., King, H. W., Li, T., Elmentaite, R., Lomakin, A., Kedlian, V., Gayoso, A., Jain, M. S., Park, J. S., Ramona, L., Tuck, E., Arutyunyan, A., Vento-Tormo, R., Gerstung, M., James, L., Stegle, O., Bayraktar, O. A.
Velten, B., Braunger, J.M., Argelaguet, R., Arnol, D., Wirbel, J., Bredikhin, D., Zeller, G., Stegle, O
Jerber, J., Seaton, D. D., Cuomo, A. S. E., Kumasaka, N., Haldane, J., Steer, J., Patel, M., Pearce, D., Andersson, M., Bonder, M. J., Mountjoy, E., Ghoussaini, M., Lancaster, M. A., Marioni, J. C., Merkle, F. T., Gaffney, D. J., & Stegle, O.
Bonder, M. J., Smail, C., Gloudemans, M. J., Frésard, L., Jakubosky, D., D’Antonio, M., Li, X., Ferraro, N. M., Carcamo-Orive, I., Mirauta, B., Seaton, D. D., Cai, N., Vakili, D., Horta, D., Zhao, C., Zastrow, D. B., Bonner, D. E., Wheeler, M. T., Kilpinen, H., … , Stegle, O.
Get in touch with us






