Translational Radiotheranostics

Dr. Martina Benesova-Schäfer
Group Head
In the 21st century, cancer will remain one of the leading causes of death worldwide. Our mission is to prevent this through the promising combination of personalized radiodiagnostics and targeted radionuclide therapy – known as radiotheranostics.

What drives us ?
At the heart of our research lies the Targeted Radionuclide Therapy (TRNT), an innovative method of fighting cancer. In TRNT, selective binding motifs transport radionuclides to designated tumor target structures such as receptors, antigens or enzymes. Compared to conventional External Beam Radiotherapy (EBRT), TRNT offers enormous potential for the treatment of metastatic tumors and is currently in focus for new, promising combination therapies.
In addition to this method, we are also investigating Targeted Alpha Therapy (TAT), which is of great interest in nuclear medicine. TAT uses alpha particles, which are characterized by decisive properties:
- A high linear energy transfer (LET; 50-230 keV/µm), which enables an increased relative biological efficacy compared to other established radionuclide therapies.
- Lower penetration depths into the tissue of 50-100 µm (approx. six cell layers), which can minimize damage to healthy tissue near the tumor.
The role of the immune system in tumor development and metastasis is critical and should not be neglected. As a result, the combination of TAT with immunotherapeutic approaches is particularly lucrative to achieve synergistic effects and improved treatment efficacy.
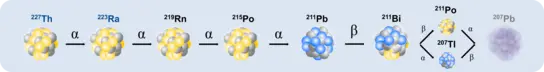
In highly innovative and interdisciplinary projects, we currently focus on alpha-particle emitting radionuclides, such as Th-227, Ac-225 and Ra-223, and investigate their coordination properties, physicochemical effects and radiation-induced immune responses in tumor tissue and the microenvironment.
We use this knowledge to identify new therapeutic targets and synthesize effective radioligands/radiopharmaceuticals for diagnostic imaging, endoradiotherapy and intra-operative surgery in order to apply our promising findings as quickly as possible for improved personalized precision medicine in oncology.
What are we currently working on?
Radionuclide Separation and Purification
We develop efficient methods for the separation and purification of α-emitters to expand their use in preclinical research and clinical applications. These strategies enable effective separation without complex subsequent steps, improved availability of α-emitters and are transferable to other clinically relevant radionuclides.
Radionuclide Generators
With novel prototypes of radionuclide generators, we want to enable the (de)centralized production of α-emitters without the need for accelerators or nuclear reactors. The immobilization of the parent nuclide with subsequent elution of the daughter nuclide in high purity represents the technical foundation. More specifically, we aim to build ²²⁷Ac/²²⁷Th and ²²⁷Th/²²³Ra-based generator systems from the reprocessing of uranium, thorium and nuclear waste in order to make relevant α-emitters more easily available for research (More here).
Structure-Function Relationships
Structure-function relationships of actinides need to be explored to predict their magnetic, electronic and (radio)chemical properties. Using high-energy X-ray absorption spectroscopy (HR-XANES) and extended X-ray absorption fine structure (EXAFS), we analyze pharmaceuticals and chelating agents in collaboration with the Karlsruhe Institute of Technology (KIT). Density functional theory (DFT) and quantum mechanics of atoms in molecules (QTAIM) help us to understand the interaction effects between metal ions and ligands.
![The image features a chemical structure diagram showing a compound labeled as [^177Lu]Lu-PSMA-617. Key elements include various nitrogen and carbon atoms arranged in a complex formation, with annotations for different isotopes represented in blue and red text.](/fileadmin/_processed_/5/4/csm_F_Alt_Mol_2af0d19c4f.webp)
Radiolabeled Substances for Diagnosis and Therapy
Novel or optimized radiolabeled ligands and pharmaceuticals are essential for the effective diagnosis and therapy of cancer. Our compound database includes small and hybrid molecules as well as peptides and peptidomimetics with shorter or longer retention time, which are obtained by classical organic or solid phase synthesis and coordination chemistry methods, which are crucial for our radiotheranostic agenda (More here).
Multifunctional Inorganic-Organic Hybrid Nanoparticles (IOH-NPs)
The recoil phenomenon in radiopharmaceuticals with α-emitters affects their stability. We aim to address this problem with the new concept of Inorganic-Organic Hybrid Nanoparticles (IOH-NPs), which is being developed by our project partners at the Karlsruhe Institute of Technology (KIT). Such nanoparticles contain functional anions that can improve pharmaceutical properties. IOH-NPs enable various advantages, such as high functional loadings, fast water-based synthesis procedures, improved reproducibility.
Functional Alternatives
We are researching the application of functional alternatives to radiometal-based drugs for in vitro development phases. We realize this e.g. through robust tritium chemistry and non-radioactive metal substitutes. We have successfully confirmed this using the isotopologue ([α,ß-³H]Nal)Lu-PSMA-617 (More here). This strategy can easily be applied to other cancer targets and is a useful alternative for scientific institutions when access to clinically relevant radiometals is not possible.
Uptake and Efflux Transporters
Membrane transport proteins in various organs have a strong influence on the absorption, distribution, metabolism and excretion (ADME) of applied (radio)pharmaceuticals. In order to optimize our drug development in a targeted manner, we investigate the underlying receptor-specific and non-specific interactions of our proprietary (radio)pharmaceuticals (More here).
Resistance Development
This project investigates critical gene expression patterns associated with treatment resistance in advanced prostate and salivary gland cancer. Our data will be obtained from blood and tissue samples from patients with advanced adenoid cystic (ACC), ductal (SDC) and mucoepidermoid (MEC) salivary gland cancers undergoing alpha-targeted therapy (TαT). The study aims to develop personalized predictive models for treatment response and to evaluate potential TαT-based treatment alternatives for ACC, SDC and MEC patients who have exhausted established treatment options.
DNA Damage and Repair
Evaluation of α-emitter-induced DNA damage and repair (DDR) modulation holds great potential for combined (synergistic) TαT with a DDR-specific agent. Flow cytometry, microscopy and protein- and DNA-based assays are used to investigate the induction and repair of double-strand breaks (DSB). Subsequently, these parameters will be validated and characterized in vivo to gain critical translational insights. We expect this combined treatment to outperform conventional approaches in efficiency and lead to improved anti-tumor effects and prolonged remission in cancer patients.
Late-stage Effects
Patients who undergo TαT often show severe and acute side effects and currently unknown or unpredictable late effects. With this study, we want to close this knowledge gap and investigate the histological, pathogenic (long-term) changes caused by TαT in mouse models. Tumor and healthy tissue exposed to α-emitters will be evaluated by pathological assessment, multiplex immunohistochemistry (mIHC) and MALDI imaging (MSI) to identify altered protein abundance. This deeper understanding of the underlying pathomechanisms should enable protective adjunctive therapies to eliminate unwanted side effects.
Our milestones
Two of our members contributed to the development of PSMA-targeted compounds (PSMA-11, PSMA-617, PSMA-1007) in the former Department of Radiopharmaceutical Chemistry of the DKFZ. PSMA-617 was licensed from ABX (DE) and acquired by Novartis (CH) for USD 2.1 billion in 2018.
2022 [¹⁷⁷Lu]Lu-PSMA-617 (Pluvicto®) received approval from the FDA, MHRA, Health Canada and EMA for the treatment of metastatic prostate cancer: a global success.Our in-house developed products [⁶⁸Ga]Ga-PSMA-11 (Locametz®) and [¹⁸F]PSMA-1007 (Radelumin®) are used worldwide for PET diagnostics of prostate cancer.
These developments show that our research contributes directly to improving patient care, with the aim of successfully applying these advancements to other types of cancer beyond prostate cancer.

Meet the team
The Junior Research Group Translational Radiotheranostics (formerly: Molecular Biology of Systemic Radiotherapy) was established in April 2019 and brings together researchers from various scientific backgrounds to advance our interdisciplinary research projects and develop innovative approaches to improve systemic radiotherapies.
-

Dr. Martina Benesova-Schäfer
Group Head
- Show profile

Sandra Casula
Administrative Assistant for Divisions E041, E270 and E280
-
Ulrike Bauder-Wüst
Biologist
-
Dr. Luciana Kovacs dos Santos
PostDoc - Translational Radiooncology
-

Dr. Adela Simkova
PostDoc - Organic Chemistry
-

Dr. Harun Tas
PostDoc - Organic & Coordination Chemistry
-

Dr. Laurène Wagner
PostDoc - Radiochemistry
-

Dr. Ruth Winter
PostDoc
Selected Publications
Schäfer M, Bauder-Wüst U, Roscher M, Motlová L, Kutilová Z, Remde Y, Klika KD, Graf J, Bařinka C, Benešová-Schäfer M*
Winter RC, Amghar M, Wacker AS, Bakos G, Taş H, Roscher M, Kelly JM, Benešová-Schäfer M*
Taş H, Bakos G, Bauder-Wüst U, Schäfer M, Remde Y, Roscher M, Benešová-Schäfer M*
Bauder-Wüst U, Schäfer M, Winter R, Remde Y, Roscher M, Breyl H, Poethko T, Tömböly C, Benešová-Schäfer M*
Get in touch with us


