Research for a life without cancer
At the DKFZ, we want to ensure that fewer people develop cancer, that cancer can be cured or treated so effectively that those affected can live with the disease and grow old with a good quality of life.
About DKFZ







Latest from DKFZ
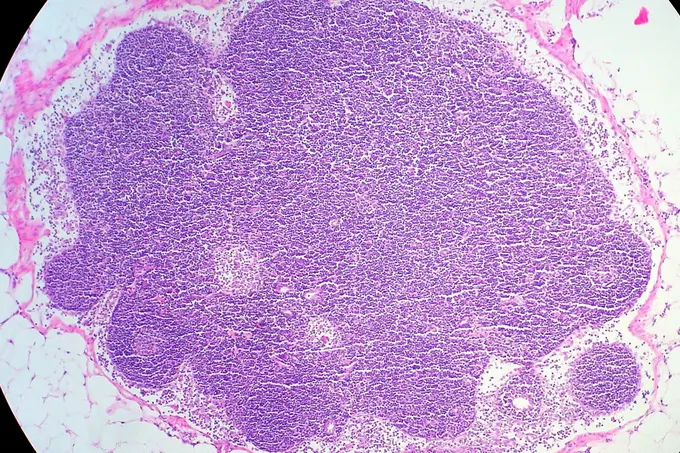
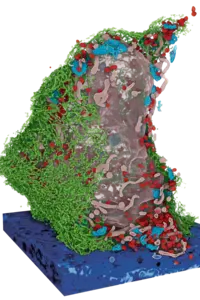
For the first time, researchers have succeeded in mapping the organization of immune cells in human lymph nodes. The study was led by scientists from Heinrich Heine University Düsseldorf, Düsseldorf University Hospital, the German Cancer Research Center (DKFZ), the European Molecular Biology Laboratory (EMBL), and the Max Delbrück Center (MDC) in Berlin. They were able to demonstrate why the architecture of healthy lymph nodes is altered in malignant lymphomas.



Our research opens doors in the fight against cancer


Do you have questions on the topic of cancer?
Let us advise you!
Doctors from the Cancer Information Service answer your questions every day. Find out more now for free!