Smart Technologies for Tumor Therapy

Prof. Dr. Tian Qiu
Department Head
Wireless micro-/nano-robots hold great potential for minimally invasive medicine. They may enable targeted drug delivery to tumors, long-term precise in vivo measurements, and they may also enable new surgical procedures at cellular level. Our group works at the interface between microsystem engineering and biomedical science, to develop new robotic hardware and software for a more precise and personalized cancer therapy.
Our Research
Micro-/nano-robots for Tumor Therapy
Drug delivery efficiency poses a significant challenge in the effective treatment of cancer. Conventional delivery methods show very low efficiency (<1%) in the long journey from the bloodstream to the tumor cells, due to several stages of biological barriers. They lead to systemic toxicity and limited drug concentrations. Despite the recent development of passive nanocarriers, their therapeutic potential is often undermined by inadequate targeting and penetration into the solid tumor tissues.
Active micro-/nano-robots offer a unique chance to tackle this challenge. Our vision is that self-propelling micro-/nano-vehicles can be controlled to navigate in the human body and penetrate solid tumor tissues, to enhance the drug uptake and improve the overall efficacy of tumor therapies. At DKFZ site Dresden, we are aiming at the holistic integration of four key aspects into one intelligent microsystem: locomotion, sensing, control and biomedical cargos. We are in close collaboration with UKDD, NCT, OncoRay, HZDR and DKTK to test the developed robots in preclinical trials to validate and optimize the technology.
Third-party funding
Our group is working on an exciting ERC starting grant project "VIBEBOT" (Vibrational Microrobots in Viscoelastic Biological Tissues) and aim at deep tissue penetration and sensing with intelligent microsystems. Micro-/nano-robotics will bring new approaches for the active and targeted delivery of therapeutics to tumors, facilitating precision oncology for future medicine.

Projects
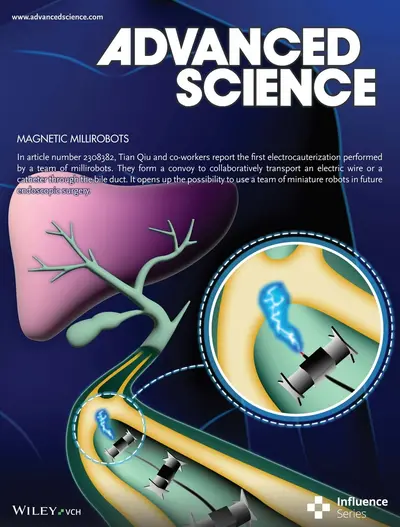
TrainBot – a team of micro-robots for endoscopic surgery
Current members: Moonkwang Jeong, Dr. Meng Zhang, Jyothi Kumari Mariyanna
Small-scale robots face challenges with locomotion on slippery biological tissues, which is essential for performing effective medical tasks. We developed a team of millirobots called TrainBot that work collaboratively to transport heavy loads through narrow biological lumens [ref]. Each robot's feet are optimized to enable effective movement on slippery surfaces. We demonstrate the first electrocauterization performed by miniaturized robots to clear a biliary obstruction for fluid drainage and drug delivery, highlighting the potential of collaborative small-scale robots for surgery.
Ref.: https://onlinelibrary.wiley.com/doi/10.1002/advs.202308382

Nanorobots for targeted delivery in deep tissues
Current members: Dr. Haoying Wang, Dr. Yue Gao, Yunfei Zhang
Key collaborator: Prof. Alexander M Leshansky, Technion, Israel
In our previous research, we developed the world's smallest nanorobot that can penetrate real biological tissues [ref]. The nanopropellers mimic the corkscrew propulsion of bacteria and have a spherical head and a helical tail with a diameter of ~500 nm, which perfectly matches the nano-sized pores of the targeted biological tissue. The propellers exhibit a finite magnetic moment in the diametric direction and when actuated under a rotating magnetic field, the rotation is coupled to translation due to its helical shape. Our experimental results show that a swarm of nanopropellers can be controlled to navigate over centimeter distance in the eye and reach the targeted region (optic disc) of ~6 mm in diameter on the retina. One key research question is whether the active nanoparticles can be wirelessly guided to penetrate solid tumor tissues and more efficiently deliver cancer drugs.

Human-scale magnetic control systems
Current members: Moonkwang Jeong, Ann-Sophia Mueller
Traditional electromagnetic actuation systems often have limited accessible volume, which restricts the controllable working space of the robots for in vivo medical applications at human scale. In this project, we develop a new magnetic actuation set-up comprised of a pair of static magnets and a rotating magnet, which generates a rotational magnetic field vector on a virtual conical surface, in a large accessible volume. The permanent magnetic actuation set-up requires neither an expensive power amplifier nor a coil cooling system, has a large enough accessible volume that can in future accommodate a human patient. The project was presented at IEEE MARSS 2023 [ref], and won the award nomination of "The Best Student Paper".
The group is developing new machine learning methods to accurately localize and precisely control the micro-robots in complex biological environments.
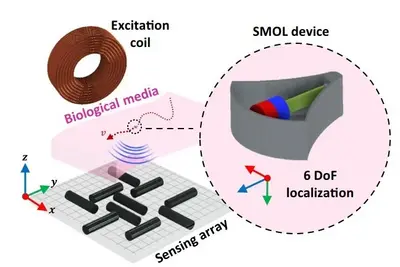
SMOL – magnetic localization of wireless devices
Current members: Felix Fischer, Dr. Liyuan Tan, Dr. Haoying Wang
Former member: Dr. Christian Gletter
One common challenge of current minimally-invasive medical instrument and implants is the reliable and wireless tracking of their positions deep in the human body. State-of-the-art imaging methods, such as ultrasound, MRI, and fluoroscopy/CT, have difficulties to continuously, wirelessly, safely and accurately monitor the three-dimensional position and orientation of microdevices. We developed small-scale magneto oscillatory localization (SMOL), which utilizes a miniature magnet attached to a cantilever to form a mechanically resonant system.Thanks to the passive design, no batteries or wires are required, and the device can be miniaturized to millimeter-scale, with large tracking depth up to 12 cm and very high accuracy and precision below 1 mm.It offers real-time spatial tracking for micro-robots as well as minimally-invasive surgical tools and medical implants.
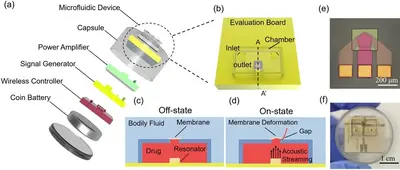
On-demand wireless drug implants
Current members: Dr. Jiyuan Tian
Former member: Dr. Yangchao Zhou
On-demand controlled drug delivery is essential for the treatment of chronic diseases. The drug is released at the time when required, thus its efficacy is boosted and the side effects are minimized. However, most state-of-the-art drug delivery methods rely on slow and uncontrollable passive diffusion. Active drug delivery methods, such as thermopneumatic pumps and electromagnetic valves were recently developed, however, they still suffer from slow response and large device size. We developed a miniaturized microfluidic device for wirelessly controlled ultrafast active drug delivery, driven by a radio-frequency oscillating solid-liquid interface. The oscillation generates acoustic streaming in the drug reservoir, which opens an elastic valve to emit the drug with very fast response of the delivery on the order of 1 ms, more than three orders of magnitude faster than the start-of-the-art.
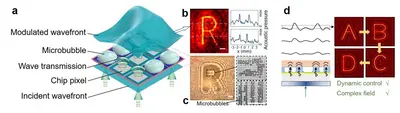
Acoustic hologram for contactless cell manipulation
Current members: Dr. Meng Zhang
Key collaborator: Prof. Zhichao Ma, Shanghai Jiaotong University, China
We developed a radically new approach that enables the generation of complex 3D ultrasound fields with much higher complexity. This work has been published in Nature. We used a 3D-printed highly-structured phase plate – the acoustic hologram – to achieve arbitrary shaped acoustic pressure fields. A plane wave coming from a single transducer that passes through the acoustic hologram element experiences a phase modulation across its wavefront. The 3D-printed acoustic hologram can be finely structured and can thus give rise to diffraction-limited arbitrary shaped acoustic pressure fields with unparalleled complexity. Recently, we also develop a new MEMS device for the dynamic modulation of an acoustic hologram, and demonstrated the capability of using ultrasound field to wirelessly manipulate soft microparticles and pattern cells.
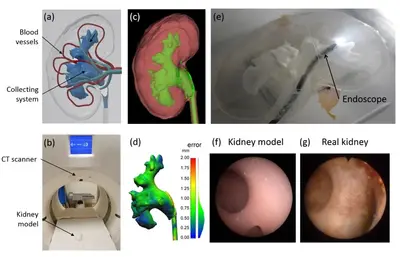
3D printing in vitro tumor models
Current members: Moonkwang Jeong, Ann-Sophia Müller
Former members: Dr. med. Xiangzhou Tan, Dr. Eunjin Choi, Nurcemal Atmaca, Dandan Li Key Collaborator: Prof. Arkadiusz Miernik, Department of Urology, University Hospital Freiburg
Additive manufacturing enables the design and fabrication of objects with complex three-dimensional shapes that cannot be obtained from standard subtractive machining approaches. The research project aims to develop the process to 3D print a soft printing material that simultaneously has high spatial resolution and high resemblance of real biological tissues. We demonstrate the range of potential applications by fabricating in vitro organ models with a resolution of tens of micrometers, including hollow branched networks that represent blood vessels, using materials, such as gels, that are soft and biocompatible. The method can generate realistic 3D complex environment with cells and extracellular matrices, which can be widely applied for in vitro experiments to test new biomedical devices and robots.
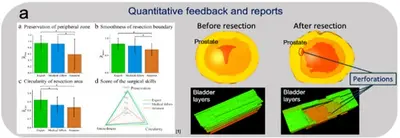
Cyber-physical organ models for surgical simulation
Current members: Ann-Sophia Müller, Moonkwang Jeong
Former member: Do Yeon Kim
Artificial Intelligence (AI) has revolutionized many fields, such as computer vision and natural language processing. These beneficial areas share the common features that the available data are abundant, standardized, accessible and relatively cheap to acquire. However, when similar machine learning approaches are applied to biomedical problems, one major challenge is how to obtain a large amount of data under realistic conditions.
We develop the digital twin of human organs that combines the advantages of physical and cyber models, to facilitate the sensing of hard-to-acquire biomedical data under realistic surgical conditions. We created a fully-automated pipeline for the quantitative outcome assessment in surgical trainings. For example, with a focus on the transurethral resection of the prostate (TURP) and the transurethral removal of bladder tumor (TURBT). A customized organ scanner and machine-learning algorithms are developed to measure the organ phantoms and pin-point mistakes, providing quantitative data to accelerate the training of surgeons and surgical robots.
Team
We are an international team forming by members from 5 different nations, and also with interdisciplinary scientific backgrounds in the fields of biomedical and microsystem engineering, materials science, computer science, chemistry and medical science. We are joining forces to develop new robotic technology to fight against cancer.
-

Prof. Dr. Tian Qiu
Head of Department
-

Nicole Türkowsky
Assistant
-

Dr. Jiyuan Tian
Postdoctoral Researcher
-

Dr. Haoying Wang
Postdoctoral Researcher
-

Dr. Yue Gao
Postdoctoral Researcher
-

Ann-Sophia Müller
PhD Student
-

Moon Kwang Jeong
PhD Student
-

Jyothi Kumari Mariyanna
PhD Student
Selected Publications
M. Jeong, X. Tan, F. Fischer, T. Qiu*
D. Wu, …, T. Qiu, P. Fischer, M. G. Shapiro et al.
F. Fischer, C. Gletter, M. Jeong, T. Qiu*
Z. Ma, …, T. Qiu*, P. Fischer*
Z. Wu, …, T. Qiu*, P. Fischer*
T. Qiu, D. Schamel, A. g. Mark, P. Fischer
News

Tian gave a presentation on “Micro-/nanorobots for Tumor Therapy” at the TREFFpunkt Gesundheitsindustrie (MEETINGpoint health industry) event in Stuttgart.
© Foto: BIOPRO Baden-Württemberg GmbH - Frank Eppler
TREFFpunkt Gesundheitsindustrie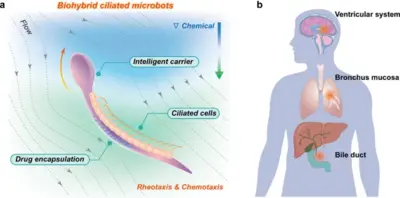
Congratulations! Our team is collaborating with Helmholtz Bioengineering on the CiliaMAT project in the field of biomedical engineering. This project will develop soft microrobots combining smart materials with living ciliated human cells, that can sense flow and chemical cues to autonomously navigate the body, target tumors, and deliver therapeutic compounds precisely.
Read more
Tian was invited to give a talk at the 3rd Expert Summit on the Future of Deep Brain Stimulation in Würzburg.
Retune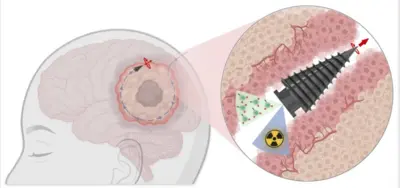
Congratulations! Our team is collaborating with Helmholtz Bioengineering on the RoboT3 project in the field of biomedical engineering. This project will create a new paradigm for minimally invasive access to the deep brain and treat hard-to-reach brain tumors using micro-robot therapeutic delivery and sensing.
Read more
We congratulate you on the successful completion of your doctoral thesis and wish you all the best for your professional and personal life! :)

Our team presented 3 papers at the IEEE International Conference on Intelligent Robots and Systems (IROS). Well done, Meng, Jiyuan and Sophia!

Again we had a great group retreat, with a lot of good presentations, conversations and fun – this time at the Kühlhaus in Görlitz. Thanks to our guests Sona Michlikova from OncoRay and Rudi Apolle from NCT for joining us to strengthen our collaboration. We are looking forward to next years retreat! :)

It was great having you here in Dresden and we wish you all the best for your future study and career!

We are very lucky to have you here in Dresden and wish you all the best for your future study and career!

Tian gave a keynote talk on “Micro-robots for minimally-invasive surgery” at CURAC 2025 in Heidelberg.

Once again our Tumor Blasters took part at the charity run with aiming 86 km in total. The run was held in aid of oncology projects at the National Center for Tumor Diseases (NCT/UCC) in Dresden. Among other things, the event supports sports therapy in pediatric oncology.

Congratulation goes to Liyuan Tan, Felix Fischer und Tian Qiu for the nomination of the Best Conference Paper Award at the International Conference on Manipulation, Automation and Robotics in USA 2025.

Congratulations to our postdoc Dr. Yue Gao for securing the Humboldt postdoctoral fellowship!

A successful “Long Night of Science” lies behind us. Our stand at the NCT Dresden was once again visited by numerous visitors. A highlight this year were a Science Slam, jointly organized by the EKFZ for Digital Health and DKFZ Patient Advisory Board Dresden, where Sophia slamed about our “Micro-heroes: What tiny robots can do for our health.” We are already looking forward to next years Science Night!

We congratulate you on the successful completion of your doctoral thesis with summa cum laude and wish you all the best for your professional and personal life! :)

Tian presented at the 4th German Cancer Research Congress in Heidelberg on May 26-27, 2025 on the topic “Wireless self-propelling micro-robots for the precise treatment of glioblastoma”.

At ICRA (International Conference on Robotics and Automation), Sophia presented our recent work on controlling millirobots by optimizing a one-sided array of permanent magnets to create a stable 2D magnetic force trap in open space at human-scale distances (20–120 mm). A GPU-accelerated optimization algorithm rapidly computes the ideal magnet orientations. A prototype with just two magnets allows us to control a millirobot along complex trajectories. We also demonstrate scalability by optimizing arrays of up to 100 magnets in under three seconds.

following
The paper “A Magnetically Transformable Twisting Milli-robot for Cargo Delivery at Low Reynolds Number” presents TwistBot, a magnetically transformable millirobot that changes its shape from a flat ribbon to a helix under a magnetic field, enabling propulsion in viscous fluids.
Read here.
Tian gave an invited talk on “Cyber-physical Organ Models for Surgical Robots and Medical Training”.

Congratulations to Dr. Jiyuan Tian on being awarded the prestigious Marie Skłodowska-Curie Postdoctoral Fellowship for his project HyBOT!

Prof. Daniel Cahill, Department of Neurosurgery, Massachusetts General Hospital and Harvard Medical School visited our division.

Our work on wireless miniature sensors gets highlighted by the Helmholtz Biomedical Engineering under the Flagship 5 - Generating smart implants.
Read here.
Our recent group retreat was a resounding success! It was an incredibly enriching and engaging experience that highlighted our great team. We learned a lot and strengthened our bonds, and we can't wait for the next years retreat! :)

Congratulations to our DKFZ/S3T - Tumor Blasters for running 38 km to benefit oncology projects, including sports therapy for children at the Nacional Center for Tumor Diseases Dresden (NCT/UCC). Well done!
… at the National Decade Against Cancer
Read here.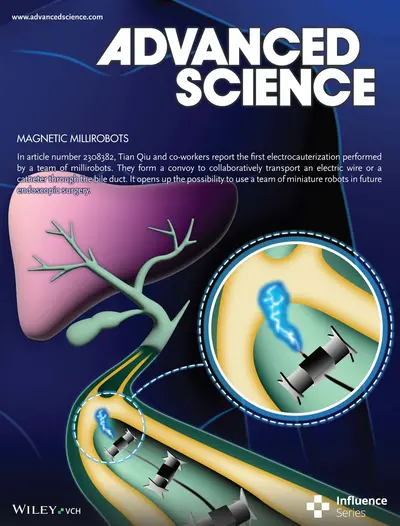
Read our paper with the title: "A Convoy of Magnetic Millirobots Transports Endoscopic Instruments for Minimally-Invasive Surgery"
Read here.
Congratulations to Ms. Gargi Thakur, a bachelor student in EE from the Indian Institute of Science Education and Research (IISER), Bhopal, for a successful 3-month internship at our division! We are very lucky to have you here in Dresden and wish you all the best for your future study and career!

… for the new building of the German Cancer Research Center in Dresden
Prof. Hao Zeng, Tampere University, visits our division and gives a talk on " Liquid Crystal Elastomers: Self-Oscillation and Flight"

Prof. Zhichao Ma, Shanghai Jiao Tong University, visits our division and gives a talk on "Contactless micro-manipulation via ultrasound and its biomedical applications".

Congratulations to Jiyuan and Felix for their successful talks at IEEE MARSS 2024, Delft, Netherlands.
Jiyuan presented "Yield stress fluid as ball joints for magnetic miniature robots".
Felix presented "Real-time tracking and control of mini-robots using magneto-oscillatory localization".
Tian and Prof. Hao Zeng, Tampere Univ, Finland co-organized a special session on "From soft materials to soft microrobots", a full-day workshop with 10 exciting talks.

In the paper "A Miniaturized Device for Ultrafast On-Demand Drug Release Based on a Gigahertz Ultrasonic Resonator" we present a miniaturized device for wirelessly-controlled on-demand drug delivery. A solid-liquid interface oscillates at gigahertz frequency to generate acoustic streaming, which opens an elastic valve and emits the drug with an ultrafast response of 1 ms. The system shows great promise as a long-term controlled drug delivery implant for chronic diseases.
Read here.
We presented our research on the actuation and localization of micro-/nano-robots. Numerous live demonstrations and presentations attracted visitors of all ages to try out, explore and marvel. It was great fun to share our research. We look forward to meeting you next year!
… on the Helmholtz homepage
Read Tians interview here.
The paper describes the precise localization of miniature robots and surgical instruments inside the body.
Read the open-access paper here.
Congratulation goes to Moonkwang Jeong for the nomination of the Best Student Paper Award at the International conference on Manipulation, Automation and Robotics at Small Scales in Abu Dhabi 2023
Vacancies
Our young, highly motivated and creative team is always open for new talents, that enjoy to work in a friendly and collaborative area.
We are looking forward to receive your application via mail to: Ms. Nicole Türkowsky nicole.tuerkowsky++at++dkfz.de including the following in a single PDF file:
- A motivational letter describing why you want to join our group. Also mention if you are interested in a specific project and how your experience/education relates to this project
- A short CV
- A recent overview of grades from your studies, include final grades if available
- A copy of your school graduation certificate
- Letters of references
Get in touch with us

