Success Stories
Vaccine against cervical cancer
HPV_1
© DKFZ/Prof. Dr. H. Zentgraf
The innovative vaccine against cervical cancer marks a milestone in the prevention of what is the second commonest cancer in the world and the third-ranking cause of death in women. More than 4,500 women a year in Germany still contract cervical cancer and around 1,500 die from the disease. If all girls were to be vaccinated in time, these figures could be drastically reduced. In 2014, the Standing Vaccination Committee at the Robert Koch Institute recommended lowering the age for HPV vaccination to between 9 and 14, so that more girls than ever can be protected from the infection before they become sexually active. Moreover, at this younger age only two vaccinations are required to afford lifelong protection. The vaccine costs are reimbursed by the health insurers.
Two vaccine products have now received market approval in over 50 countries: Gardasil® from Merck & Co and Cervarix® from GlaxoSmithKline (GSK). Together they produce annual sales in the billions. Both vaccines provide almost complete cover against HPV types 16 and 18, which are responsible for around 70 percent of all cases of cervical cancer. Since Gardasil is also targeted against HPV 6 and HPV 11, which cause other genital disorders such as genital warts (condylomata), approximately 90 percent of these cases can be prevented by vaccination. A successor product, Gardasil 9, has now been approved by the US and European regulatory authorities and has now been available on the German market since 2016. In addition to HPV types 6, 11, 16 and 18, Gardasil 9 is also targeted against five other types (31, 33, 45, 52 and 58), which account for around 20 percent of all cases of cervical cancer.
Links:
https://www.krebsinformationsdienst.de/vorbeugung/risiken/hpv-impfung.php
High-precision radiosurgery of tumors

CyberKnife® / iris collimator
© DKFZ/Marco Müller
"Accuray was a logical choice for us in selecting a research partner given our longstanding collaboration on the CyberKnife® System and the company's leadership in imaging systems and motion management", said Professor Wolfgang Schlegel on the conclusion of the collaboration agreement with the American company. The joint projects also include, in close cooperation with Heidelberg University Hospital, researching and expanding intensity-modulated radiation therapy (IMRT) and the treatment options for Tomotherapy® - an Accuray technology for reducing the exposure to side effects during the radiotherapy of tumor patients.
Links:
Breaking the diffraction barrier
© DKFZ/Abt. Optische Nanoskopie
In the "Optical Nanoscopy" Laboratory at the DKFZ and in combination with an ultrafast electro-optical scanner developed by the engineer Dr. Jale Schneider at RWTH Aachen University - an STED nanoscope has been developed, under the direction of Stefan Hell and Johann Engelhardt, that enables images to be recorded in intervals of milliseconds. This is several thousand times faster than has previously been possible with laser scanning microscopes. As a result, dynamic processes within the cell that had not been accessible to optical analysis to date can now be examined in detail.
Links:
http://www.nobelprize.org/nobel_prizes/chemistry/laureates/2014/hell-lecture.html
Tetravalent bifunctional antibodies

TandAb® model
© Affimed 2014
Affimed had in-licensed the TandAb® technology platform from the DKFZ. Two of the antibody-like anticancer substances are in clinical development: AFM13 for treating Hodgkin lymphoma in clinical phase 2 and AFM11 for non-Hodgkin lymphoma in phase 1; other immune molecules are still at the preclinical testing stage. Affimed has entered into collaboration with Merck/MSD to test combination therapies for Hodgkin lymphomas with AFM 13.
Following a successful IPO in September 2014, Affimed Therapeutics BV (AFMD for short) is now listed on NASDAQ, the American technology stock exchange. Analysts report that Affimed has good prospects for further share price rises. For the DKFZ this represents the second successful exit of a spin-off. The DKFZ profits both from the growth in the value of the shares, which can be sold at a later date, and from licenses through product sales. Affimed invests its income from the IPO primarily in the future development of its innovative antibody programs.
Links:
http://www.affimed.com/pdf/160125_afmd_merck_release_final.pdf
Active substance against the most malignant brain tumor

A glioblastoma in the MRT
© DKFZ/Med. Physik in der Radiologie, Foto: Michael Bock
In preclinical tests, Apogenix was able to show that APG101 restores the formation of blood cells (erythropoiesis) in patients suffering from myelodysplastic syndromes (MDS). MDS is an anemic bone marrow disorder associated with the risk of developing into an acute myeloid leukemia, a cancer of the blood. Apogenix then conducted a phase I clinical trial with APG101 in the treatment of transfusion-dependent MDS patients with severely impaired erythropoiesis in order to test the safety, efficacy and tolerability of the drug. The results of the study are expected in the summer of 2016.
APG101 possesses Orphan Drug Status for the treatment of gliomas in the EU and for the treatment of glioblastomas and MDS in the USA. The company CANbridge Life Sciences (Beijing, China) is responsible for the licensing and marketing of APG101 in China, Macao, Hong Kong and Taiwan. Thanks to this partnership, Apogenix can earn revenues that can then be used, in turn, for its own development in the other countries.
Links:
http://apogenix.com/files/pdfs/DE/20160615_Apogenix_MDS_Topline_Data_DE.pdf
Software for improved diagnosis and therapy planning
© Mint Medical GmbH
The company has introduced a certified quality management system and applies this to its products, which are both compliant with medical devices legislation and CE-tested. "Mint Liver" was developed as a tool specifically for the computer-aided diagnosis and therapy planning of liver diseases. It can provide a 3D reconstruction of a patient's liver, simulate various surgical procedures and help determine the optimal strategy for the surgical procedure even before the operation and, if necessary, enable the strategy to be quickly adapted to new findings during the operation.
The software development mint LesionTM facilitates the efficient monitoring and follow-up of cancer treatments. As well as providing quantitative and qualitative statements on the development of tumors during the course of treatment, Lesion can also be used to produce assessments according to the established standards in radiology simply, quickly and flexibly. Mint Medical products are now used worldwide in routine clinical practice for cancer screening, cancer staging and for assessing the response to cancer treatments. During clinical trials, LesionTM supports the reporting process in accordance with the criteria. It can be individually configured for case-specific, multicenter clinical trials. As a Clinical Trial Management System that has been approved by the FDA, mint LesionTM can support project management, study-compliant read procedures and data management in the implementation of clinical trials for imaging CROs and pharmaceutical companies. For imaging biomarker analysis, mint LesionTM can cover the whole workflow, including all the associated tasks of imaging biomarker research.
The CEO of Mint Medical GmbH, Dr. Matthias Baumhauer describes the company's vision as follows: "Mint Medical products assign a central role to the patient as an individual in our healthcare system. We therefore believe that our solutions will be an important component of any advanced hospital in the medium term."
Links:
Oncolytic viruses to treat brain tumors
Computer simulation of a parovirus
© DKFZ / Antonio Marchini
Since the preclinical studies needed to progress the project through to clinical use are very time-consuming, a partner was sought and eventually found in the company Oryx GmbH & Co KG. Oryx is specialized in taking research projects in cancer medicine through the preclinical and early clinical development stages and selling them to the pharmaceutical industry. In January 2008, the Munich-based company signed a cooperation agreement with the DKFZ and Heidelberg University Hospital, which is also involved in the development of viral therapy. Working together with industrial partners, Oryx coordinated the large-scale technical production and subsequent pharmacology and toxicology testing of the therapeutic viruses as well as the approval procedure with the Paul Ehrlich Institute. Clinical phase I/IIa trials began in the Fall of 2011 the first time that brain tumors were treated with viruses in Europe. On 12 June 2015, Oryx announced the successful completion of the clinical phase I/IIa trial on the treatment of patients with progressive primary and recurrent glioblastoma with H1 parvoviruses, in which the vaccine was found to be safe. The oncolytic parvoviruses will now be tested on other malignant tumors, including pancreatic cancers.
Links:
https://www.dkfz.de/de/presse/pressemitteilungen/2011/dkfz-pm-11-56-Mit-Viren-gegen-Hirntumoren.php
Early detection of cervical cancer
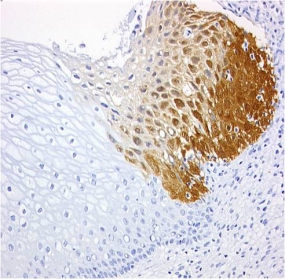
CINtec® p16 histology of a zervix carcinoma
© mtm-laboratories
In 2011, the company mtm laboratories was sold, for a total of 190 million euros, to Ventana Inc., a subsidiary of Roche, the global pharmaceutical and diagnostics group. Following the takeover, Ventana developed another product based on this technology platform, the CINtec® p16 Histology Test. As part of a fully automated immunohistochemistry assay, this enables the doctor not only to detect transformed cells, but also to precisely assign them to risk categories, an extremely important factor in the therapeutic decision.
Links:
Cell type diagnosis of metastases using antibodies to cytoskeletal proteins
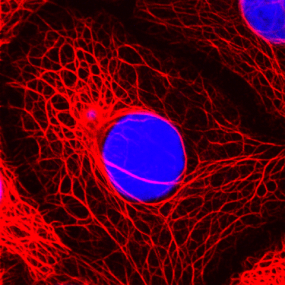
The cytokeratin skeleton of breast cancer cells
© DKFZ / Lutz Langbein
In order to meet the worldwide demand for antibodies to cell type-specific forms of intermediate filaments for use in diagnosis and biomedical research, in 1983 Franke, together with the DKFZ immunologist Professor Günter Hämmerling and two other colleagues from Heidelberg University, formed the company Progen Biotechnik GmbH, one of the very first biotech companies in Germany. The specific antibodies were further developed by Progen to produce test kits for the diagnosis of cancer and other diseases, for example rheumatoid arthritis. Immunodiagnosis with antibodies to cytoskeletal and desmosomal proteins has now become an integral part of every molecular pathology laboratory. Progen Biotechnik GmbH was wholly taken over in October 2012 by R-Biopharm AG, under whose umbrella PROGEN continued to supply the existing market and break into new markets.
Links:
http://www.dkfz.de/de/helmholtz-zellbiologie/Franke-in-Cancer_Research_DKFZ_2014_web.pdf
A radionuclide-coupled substance for the diagnosis and treatment of prostate cancer
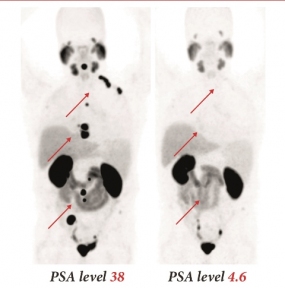
PSMA-617 mark of a prostate cancer patient
© DKFZ / Matthias Eder
Alternatively, PSMA-617 can also be bound to highly radioactive radionuclides such as lutetium-177 or actinium-225. The radiopharmaceutical is selectively taken up by the tumor cells that carry the PSMA and then destroys these cells from the inside. This could be a very promising treatment option, particularly for patients with hormone-resistant prostate cancers, which are very difficult to treat. At Heidelberg University Hospital, the team led by nuclear medicine specialist Professor Uwe Haberkorn has already used lutetium-177-labeled PSMA-617 to treat individual patients with advanced prostate cancer. After treatment with the lutetium-labeled radiopharmaceutical, levels of the prostate cancer marker PSA fell sharply in around 70 percent of the patients. PET/CT images also confirmed that metastases had shrunk or were no longer detectable. "The results were so promising that we plan to go ahead with a clinical trial as soon as possible to examine whether PSMA-617 is superior to other therapy methods," explained Haberkorn. Although other agents that target the protein PSMA and can be coupled with strong or weak radionuclides are already being developed, they have not proved to be ideal. "They are too unstable, accumulate insufficiently in cancer cells and wash out too slowly from healthy organs," explained the chemist Professor Klaus Kopka, Head of the Division of Radiopharmaceutical Chemistry at the DKFZ. "By contrast, PSMA-617 accumulates in large quantities in tumors and metastases and is stored well in cancer cells."
In June 2015, Matthias Eder, radiochemists Martina Beneová, Klaus Kopka, Uwe Haberkorn and their co-workers received the "Image of the Year Award" and the Berson-Yalow Award at the Annual Meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI) in Baltimore, USA, for their development of PSMA-617.
Links:
Antibody microarrays for screening of biomarkers in cancer

© Sciomics GmbH
In 2013, Hoheisel and his colleague Dr. Christoph Schröder, formed the biotech company Sciomics GmbH in Heidelberg, which uses its protein microarrays to offer services for medical research, diagnosis and industry including, in particular, biomarker screening, the verification of biomarker candidates, the analysis and localization of drug targets and their signaling pathways and the characterization of antibodies. Schröder is the CEO of Sciomics GmbH, while Hoheisel still acts as a consultant for the company.
The research team at the DKFZ headed by Hoheisel and Schröder has used microarrays from cancer-relevant antibodies to identify, file for patenting and publish a series of biomarker signatures. These microarrays may be important for the diagnosis, prognosis and prediction of cancer. Predictive biomarkers provide information about the possible effect of the therapeutic intervention, whereas prognostic biomarkers deliver information about the patient's illness and its course independently of the treatment. Among other aspects, they are investigating the altered plasma protein composition in patients with chronic lymphatic leukemia and other B-cell lymphomas, as well as cancer-associated proteins in pancreatic carcinoma, a type of cancer which, to date, has proved difficult to treat and usually diagnosable too late. In another study, the scientists have used an antibody microarray to predict recurrences of bladder cancer, which reappears within five years of surgical removal of the tumor in around 60 percent of patients. In their comparison of patients with and without recurrence, they have identified 255 proteins that are present in the tumor in small or large quantities. Twenty of these proteins produced a pattern which, with 100 percent specificity and 80 percent sensitivity, represents a highly promising candidate as a predictive biomarker signature for relapses of bladder cancer.
Links:
Strategic partnership with Bayer Pharma to develop innovative cancer drugs
Tumor sample, imbedded in paraffin
© Bayer HealthCare AG
In 2013, the alliance was expanded to include the field of immuno-oncology, and the corresponding researchers at BHC and the DKFZ are working together on more advanced projects in a Joint Immunotherapeutics Lab in Heidelberg. Up to 2.5 million euros a year is being invested in these projects. In total, the two partners will be investing up to six million euros a year in the collaborative oncology research projects for the period 2014 to 2018. If any projects prove successful, the DKFZ will receive a share of the revenues. Of the more than 30 projects initiated to date, many have already reached important milestones and progressed to the next phase of drug development, namely compound screening to identify new potential drug candidates.
Links:
Competency cluster in imaging and radiotherapy: the alliance with Siemens Healthcare
7 Tesla magnetic resonance tomograph
© DKFZ / Tobias Schwerdt
Links:
http://www.healthcare.siemens.de/news-and-events/prostata-krebs
Detecting and combating cancer stem cells
Tumor stem cells of pancreas cancer
© DKFZ / A. Trumpp
The scientists discovered how the loss of a tumor suppressor gene in healthy blood stem cells leads to leukemia-forming tumor stem cells. Unlike most cancer cells, tumor cells reside in protected niches and only rarely divide. As a result, they are often resistant to conventional cancer treatment. Trumpp and his team showed how dormant stem cells can be activated by chemical messengers, making them susceptible to subsequent chemotherapy. They also provided experimental evidence indicating that the tumor cells circulating in the bloodstream of breast cancer patients are accompanied by metastasis-inducing stem cells (MICs) characterized by the combination of three surface molecules: CD44, which helps cells settle in the bone marrow; CD47, which protects cells from attacks by the immune system, and MET, which enhances the cells' invasive capability. Patients with a large number of such triple-positive cells have particularly high numbers of metastases and a very poor prognosis. The characterization of the MICs leads to improved diagnosis. In cooperation with the pharmaceutical industry, substances that selectively attack the MICs, potentially improving the patient's chances of survival, are already being tested.
Links:
High-precision analyses for human gene therapy and immunotherapy
T-cell-receptor
© GeneWerk
The analysis platforms offered by GeneWerk include the following: investigation of vector safety in gene therapy through integration site analysis with (nr)LAM-PCR and target enrichment sequencing; qPCR for the simultaneous amplification and quantification of DNA or RNA fragments; immune-repertoire analyses involving the sequencing of the hypervariable region of T-cell receptors to determine the clonal and functional status of a T-cell population; (off)-target analyses for genome editing, i.e. the analysis of nonspecific genome sequence modifications outside the target site using designer nucleases (e.g. zinc finger nuclease, TALEN and CRISP/Cas 9); specific bioinformatics and data management programs for next-generation sequencing projects, whole-genome analyses, modeling and computer simulations.
Gene therapy and immunotherapy are considered to be the great beacons of hope for the treatment of hitherto incurable diseases. Major international biopharmaceutical companies are also becoming increasingly involved in these areas. With its wide range of validated analyses in its pipeline, GeneWerk is targeting this growth market. To satisfy the corresponding requirements, and barely two years after its formation, GeneWerk is pressing ahead with company certification and reorganizing the IT infrastructure so that it is able to efficiently analyze the huge amounts of generated data.
Links:
