Anogenital cancer: How cancer cells benefit from the absence of a signaling molecule
An infection with human papillomaviruses (HPV) can cause not only cervical cancer but also other types of anogenital cancer. The team of scientists headed by Professor Frank Rösl from the German Cancer Research Center (DKFZ) has now discovered that these viruses escape from immune surveillance by inactivating an important signaling molecule involved in immune responses. This hitherto unknown mechanism may yield new approaches in cancer medicine. Moreover, the molecule might also serve as a marker for detecting HPV infections before a malignant tumor develops.
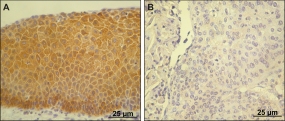
A) Normal cervical tissue containing high levels of pro-IL1β (stained brown)B) Cervical cancer, negative for pro-IL-1βPicture source: DKFZ/Charité Berlin
About ten percent of the 170 known types of human papillomavirus are considered to be carcinogenic. HPV types 16 and 18 are particularly aggressive and belong to this group. The viruses preferentially infect cells called keratinocytes which are found in epidermal and mucosal tissues. Like herpes viruses, HPV can persist in the body for a long time. Infected cells may turn cancerous many years after the initial infection.
The project of Professor Frank Rösl and his DKFZ co-worker Dr. Bladimiro Rincon-Orozco aimed at determining how HPV manages to escape surveillance by the immune system. They focused their attention on an important immune modulator called interleukin-1beta (IL-1β). “In healthy cells, this modulator is present as an inactive precursor called pro-IL-1β. When a virus invades the cell, the precursor is cleaved by an enzyme into its biologically active form and released,” Rösl explains. “IL-1β attracts immune cells that finally eliminate virus-infected cells in a multistep process”.
In cell culture experiments, the researchers discovered that a protein called E6, brought into cells by HPV, interferes with the process of pro-IL-1β maturation. Bladimiro Rincon-Orozco and the PhD student Martina Niebler infected human keratinocytes with HPV-16 or HPV-18. These cells turned out to be no longer capable of releasing mature IL-1β. The scientists discovered that this was mediated by a cellular “recycling factory” called proteasome, which normally breaks down damaged or no longer necessary proteins. E6 triggers the proteasome to eliminate pro-IL-1β. “Degradation of the precursor molecule happens at a very early stage of the infection process. This explains why cells infected with HPV-16 or HPV-18 stop producing IL-1β”, says Rösl. “If this signal mediator is missing, the immune system has difficulty with spotting and controlling the infection”.
The investigators have shown that IL-1β levels are not only decreased in HPV-infected cells under laboratory conditions, but also in patients. In collaboration with the Charité Berlin, they studied cervical cancer tissue samples from patients testing positive for HPV-16 or HPV-18. “The more advanced the cancer, the less IL-1β is produced,” says Bladimiro Rincon-Orozco. “Together with the Charité Berlin and Heidelberg University (“Kopfklinik”), we will now investigate whether these changes in the concentration of IL-1β can be used as a marker for anogenital cancer and other HPV-induced cancers.” These include tumors of the oropharynx, which are also linked to HPV infection.
This mechanism might also be useful for therapy: “So-called proteasome inhibitors are currently being tested in clinical trials,” says Rösl. These chemical substances block the proteasome, rendering it incapable of breaking down proteins. The scientists hypothesize that if cells preserve a functional form of IL-1β, they might restore the immune system’s capability to identify and eliminate developing cancer cells.
Martina Niebler, Xu Qian, Daniela Höfler, Vlada Kogosov, Jittranan Kaewprag, Andreas M. Kaufmann, Regina Ly, Gerd Böhmer, Rainer Zawatzky, Frank Rösl, Bladimiro Rincon-Orozco. Post-Translational Control of IL-1β via the Human Papillomavirus Type 16 E6 Oncoprotein: A Novel Mechanism of Innate Immune Escape Mediated by the E3-Ubiquitin Ligase E6-AP and p53. PLOS Pathogens. 2013 Aug 1. DOI: 10.1371/journal.ppat.1003536
A picture for this press release is available at:
www.dkfz.de/de/presse/pressemitteilungen/2013/images/Normalgewebe_und_Gebaermutterhalskrebs.jpg
Caption:
A) Normal cervical tissue containing high levels of pro-IL1β (stained brown)
B) Cervical cancer, negative for pro-IL-1β
Picture source:
DKFZ/Charité Berlin
With more than 3,000 employees, the German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) is Germanys largest biomedical research institute. DKFZ scientists identify cancer risk factors, investigate how cancer progresses and develop new cancer prevention strategies. They are also developing new methods to diagnose tumors more precisely and treat cancer patients more successfully. The DKFZ's Cancer Information Service (KID) provides patients, interested citizens and experts with individual answers to questions relating to cancer.
To transfer promising approaches from cancer research to the clinic and thus improve the prognosis of cancer patients, the DKFZ cooperates with excellent research institutions and university hospitals throughout Germany:
The DKFZ is 90 percent financed by the Federal Ministry of Education and Research and 10 percent by the state of Baden-Württemberg. The DKFZ is a member of the Helmholtz Association of German Research Centers.

