Division of Personalized Medical Oncology
Prof. Dr. Dr. Sonja Loges
Recent improvements in specific targeting of genomic and immunosuppressive cancer drivers have led to major advances in the treatment of malignant tumors. Personalized oncology treats patients according to these specific disease mechanisms of their individual tumors. This strategy has proven successful in some cancer entities including melanoma, breast cancer and non-small cell lung cancer (NSCLC) yielding unprecedented survival even in advanced disease. However, the majority of patients still do not reach long-term survival because they eventually develop resistance mainly due to complex and heterogenous escape mechanisms evolving under treatment pressure.
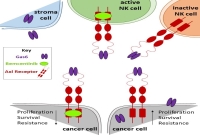
Our group identifies novel mechanisms driving cancer progression and therapy resistance with a focus on the tumor microenvironment. From these functional insights we derive novel targets and identify treatments for individualized cancer therapies. After preclinical validation we translate them into clinical studies. For example, we found that the interaction of AML cells with the bone marrow stroma elicits upregulation of its ligand Gas6 driving therapy resistance of AML cells and immunosuppression via engaging its receptor Axl in AML or immune cells, respectively (Ben-Batalla et al, Blood 2013). This pathway can be inhibited by the small molecule Axl inhibitor Bemcentinib which we translated into a clinical trial in relapsed AML patients (BGBC003, NCT02488408). In a reverse translation program attached to the trial we are dissecting patterns of response and resistance with innovative single cell sequencing technologies in order to inform further development of Axl-targeting.
In addition, we investigate patterns of response and resistance to targeted and immune therapies in cancer with a focus on precision oncology and NSCLC by utilizing innovative solid & liquid biopsy technologies. Here, we apply microfluidics techniques as well as single cell sequencing and -imaging technologies to enrich & analyze tumor cell as well as other cellular and subcellular components present in tumors and/or body fluids of cancer patients. Thereby, we identify unknown potential resistance mechanisms which we investigate in-depth at the functional level in cellular and animal cancer models concerning their capability to drive cancer progression and therapy resistance. Potentially druggable candidates are subsequently translated to clinical trials.
Overall, we aim to discover novel tumor-promoting mechanisms at the interface between tumor and host, derive novel therapies and dissect mechanisms of response and resistance in cancer patients.