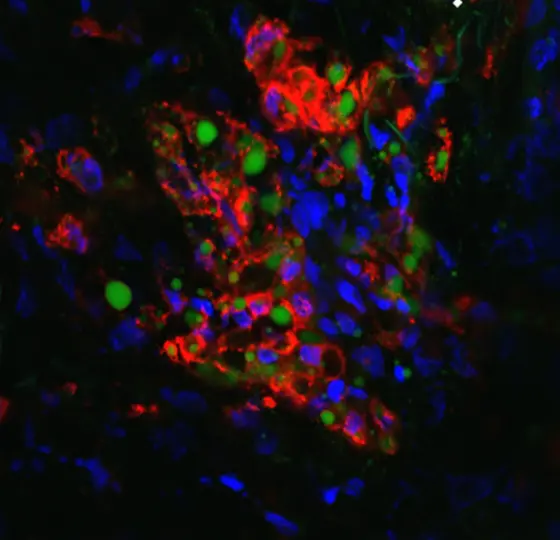
Ovarian cancer and other tumor diseases often spread to the so-called omentum—a fatty tissue layer in the abdominal cavity that covers the internal organs and does not always simply correspond to the term “belly fat.” The HI-TRON team led by Niels Halama investigated how this unique environment influences the interaction between tumor cells and immune cells.
Their findings revealed that a particularly high number of immune cells, including T cells and so-called tumor-associated macrophages, accumulate near tumor metastases in the fatty tissue. While T cells generally remain functional within the fatty tissue, macrophages experience oxidative stress due to the uptake of large amounts of fat. In this state, they contribute to the suppression of the immune response and thus promote tumor growth.
Blocking specific signaling pathways reactivates the immune defense
The researchers demonstrated that this immune-suppressive state of the macrophages can be reversed through targeted interventions.
In experiments with tumor tissue from patients as well as in mouse models, inhibiting the signaling pathway via the CCR5 receptor—for example, with the drug Maraviroc—led to the reprogramming of macrophages. As a result, T cells were once again able to penetrate the tumor more effectively and combat cancer cells more efficiently. The already approved drug Maraviroc was originally developed for the treatment of HIV infections.
The researchers also observed a similar effect when another inflammatory and lipid transport mechanism involving the CD36 receptor was blocked. Both approaches reduced the lipid burden on the macrophages and strengthened the inflammatory immune response against the tumor.
Which patients benefit most from immunotherapy?
Furthermore, the study provides insights into which patients might particularly benefit from therapies with immune checkpoint inhibitors. An analysis of clinical data revealed that adipose tissue plays a dual role: patients with metastases in the fat-rich omentum responded more frequently to checkpoint inhibitors than women whose tumors had not spread to this tissue.
Using imaging techniques and machine learning, the researchers were even able to develop a model that can estimate the presence of omental metastases—and thus the likelihood of a response to therapy—based on CT scans.
“The results clearly demonstrate how strongly metabolic processes in the tumor microenvironment influence the behavior of immune cells. This influence brings both advantages and disadvantages for patients. Our findings may help us therapeutically influence these processes in cancer patients in the future,” says study lead Niels Halama. In the long term, the researchers expect that combination therapies—which combine immune checkpoint inhibitors with agents targeting specific metabolic or inflammatory signals—could improve the treatment of ovarian cancer and other tumor diseases. Diet and exercise could also have a beneficial effect—however, study results on this are not yet available.
Publication:
Meggy Suarez-Carmona et al.: Harnessing lipid-driven immunometabolic pathways in omental metastases to enhance immunotherapy in patients with ovarian cancer
Signal Transduction and Targeted Therapy 2026, https://rdcu.be/e6Ily
* HI-TRON Mainz was founded in 2018 as a joint venture of the German Cancer Research Center (DKFZ) in cooperation with TRON gGmbH, Mainz University Medical Center, and Johannes Gutenberg University Mainz (JGU). The institute combines the internationally renowned expertise of these partners in the field of personalized immunotherapy in Mainz with the comprehensive spectrum of cutting-edge cancer research at the DKFZ.