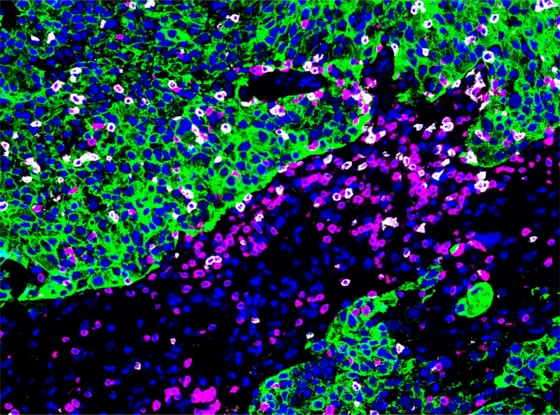
When cancer cells detach from the original tumor in the breast and form metastases in the brain, treatment options for affected patients have been limited so far. Brain metastases are more frequently associated with a poorer prognosis than metastases in other organs of the body. There is therefore a great need to better understand the cellular interactions in brain metastases and, building on this, to find a therapy that is as targeted as possible. In the newly published study, the Heidelberg researchers investigated the cellular environment of brain metastases in a total of 156 breast cancer patients, focusing on immune cells and other cells of the tumor microenvironment. To ensure the most detailed analysis possible, they combined several methods to characterize the cell types and their immune responsiveness. Through spatial analysis, the research team was also able to determine where in the tissue many immune cells ready to fight the cancer were located.
Two “immune landscapes” are associated with a more favorable prognosis
The researchers discovered patterns in the spatial distribution of immune cells that they described as “immune landscapes.” For example, if there were only a few immune cells fighting cancer around metastasis, this pattern was associated with a poor prognosis. In addition, the team identified two immune landscapes that were associated with a more favorable outcome for the patients. One of these two patterns featured a high number of tissue-resident memory T cells. These specialized immune cells presumably remain in the tissue, appear to recognize tumor cells again, and can react particularly quickly. The second favorable pattern was characterized by organized clusters of immune cells in the tumor tissue that function similarly to small lymph nodes and can support an immune response against the tumor.
Patient cells in culture confirm the important role of T cells in tumor control
“What’s special is that we were able to describe not only these patterns but also their biological function,” said Lena Jassowicz, the study’s first author. To experimentally verify the observations from metastatic tissue, the team used patient-specific models. In this process, isolated tumor and immune cells from a human are brought together in the laboratory to study their interaction directly and without interfering influences. It was found that the identified tissue-resident T cells very effectively attacked and destroyed the tumor cells of the respective patient. “We conclude from these results that these cells do indeed play an important role in tumor control,” says Fangyoumin Feng, DKFZ, also a co-first author.
These T cells could serve as a starting point for targeted immunotherapy. Immunotherapies enhance the body’s own immune response against cancer cells. However, not all patients benefit equally from such treatment. Therefore, markers are needed to predict for whom such treatment might be beneficial. The study provides important insights in this regard: In supplementary laboratory experiments, the researchers were also able to show that T-cell activity could be further increased by an immune checkpoint blockade.
Gene signatures as markers of immune landscapes
In a further step, the researchers derived characteristic patterns of active genes from their results that characterize the previously identified favorable immune landscapes. In tests using external datasets from breast cancer patients, these gene signatures reliably indicated better prognosis. Such signatures could be used in the future to better classify tumors or metastases at the molecular level and to support treatment decisions. It may also be helpful to identify immunologically “cold” metastases that are unlikely to respond to immunotherapy. This could prevent unnecessary stress caused by treatments with little prospect of success. “Simply knowing that a patient is more likely to benefit from a treatment other than immunotherapy can be clinically very valuable,” says Martina Seiffert of the DKFZ.
“This work not only provides new insights into the biology of breast cancer brain metastases but also concrete clues for the further development of personalized therapies,” says Sandro Krieg, Medical Director of the University Clinic for Neurosurgery in Heidelberg. A possible next step would be to test therapeutic strategies in clinical trials to specifically further enhance the existing immune responses against the tumor cells. “Our data clearly show that it is worthwhile to consider brain metastases as a distinct biological entity,” says Christel Herold-Mende of Heidelberg University and the UKHD. “Only by understanding these specific characteristics can we make therapies more precise and, hopefully, more effective,” says Herold-Mende.
Source: UKHD press release
Lena Jassowicz, Fangyoumin Feng, Rolf Warta, Fatima Heinicke, Amélie Wöllner, Hannah Briesch, Furkan Sor, Julia Soller, Catharina Lotsch, John K.L. Wong, Chang-Gyun Park, Nicolas Aubert, Maximilian Scheer, Zhaoqing Hu, Maximilian Knoll, Julian Schlegel, Lukas Frank, Philip Dao Trong, Gerhard Jungwirth, Amin I. Nohman, Kendra Maaß, Niels Grabe, Mandy Barthel, Simone Butz, Christina Lulay, Manuela Zucknick, Michele Bortolomeazzi, Pooja Sant, Verena Thewes, Laura Michel, Carlo Fremd, Andreas Schneeweiss, Chong Sun, Karsten Rippe, Amir Abdollahi, Andreas von Deimling, Andreas Unterberg, Sandro M. Krieg, Jan-Philipp Mallm, Peter Lichter, Marc Zapatka, Martina Seiffert, Christel Herold-Mende: Dissecting the cellular architecture of breast cancer brain metastases reveals prognostically distinct immune landscapes
Cancer Cell, 2026, DOI: 10.1016/j.ccell.2026.03.016
https://doi.org/10.1016/j.ccell.2026.03.016.