Once a tumor has reached a certain size, the cancer cells face a particular challenge: Growing in a disorganized way, tumors lack blood vessels, which leads to oxygen and nutrient deficiency inside the tumor. “Cancer cells have to completely reorganize their entire metabolism to adapt to this deficiency,“ Almut Schulze, an expert on metabolism at DKFZ, explained.
In her current work, Schulze investigated indications that the important control protein and tumor suppressor p53 plays a crucial role in adapting to deficiencies. Schulze and her team examined p53-deficient colon cancer cells to find out how p53 actually impacts on metabolic adaptation.
In order to mimic the deficient environment inside a cancerous tumor in a Petri dish, the researchers let the cancer cells grow into small spheroids. Colon cancer cells with intact p53 adapted to the deficient environment by restricting their growth. In contrast, the p53-deficient tumor cells inside the spheroids continued to grow as before, activating a metabolic pathway known as the mevalonate pathway, which supplied the cells with cholesterol, an important cell membrane component.
The mevalonate pathway can be inhibited using statins, which are common cholesterol-lowering agents. Treatment with statins did in fact induce apoptosis in the deficient environment inside the p53-deficient microtumors. In contrast, microtumors with intact p53 did not respond to the treatment.
Schulze and her team also found another way in which p53-deficient colon cancer cells adapted to the deficiencies: The activated mevalonate pathway also promoted the synthesis of ubiquinone, an important component in electron transport in the respiratory chain. In deficient environments, cancer cells are more heavily dependent on this substance – also called coenzyme Q10 – to synthesize new DNA.
“Cancer cells that are deficient in p53 use the mevalonate pathway to promote cell respiration and to access key cell components, hence ensuring their survival. From an oncologist's viewpoint, it is particularly interesting that this metabolic pathway is easy to inhibit with statins, allowing us to use a common drug to induce apoptosis in the cancer cells,“ Almut Schulze explained. “This only applies under the particular conditions of deficiency that prevail inside a tumor, however,“ she added. “That means that statins should be combined with agents that promote this metabolic stress, such as drugs that inhibit the formation of new blood vessels.“ Schulze hopes to test this new treatment strategy on tumor cells and in animal experiments as soon as possible.
Irem Kaymak, Carina R. Maier, Werner Schmitz, Andrew,D. Campbell, Beatrice Dankworth, Carsten P. Ade, Susanne Walz, Madelon Paauwe, Charis Kalogirou, Hecham Marouf, Mathias T. Rosenfeldt, David M. Gay, Grace H. McGregor, Owen J. Sansom and Almut Schulze: Mevalonate pathway provides ubiquinone to 1 maintain pyrimidine synthesis and survival in p53-deficient cancer cells exposed to metabolic stress.
Cancer Research 2019, DOI: 10.1158/0008-5472.CAN-19-0650
A picture is available for download:
Cover-pic_Composite.png
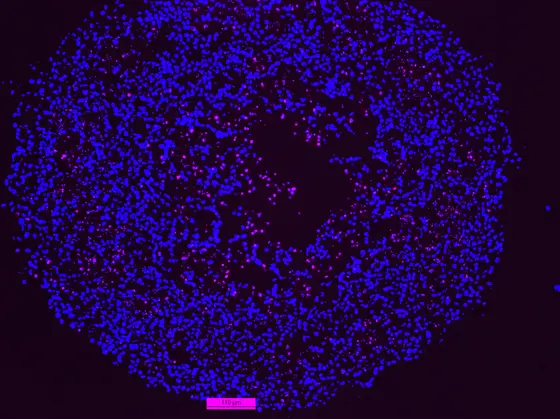
Picture Caption: p53 deficient cancer cells within the spheroids die from apoptosis (pink)
Note on use of images related to press releases
Use is free of charge. The German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) permits one-time use in the context of reporting about the topic covered in the press release. Images have to be cited as follows: “Source: DKFZ/ Almut Schulze“.
Distribution of images to third parties is not permitted unless prior consent has been obtained from DKFZ's Press Office (phone: ++49-(0)6221 42 2854, E-mail: presse@dkfz.de). Any commercial use is prohibited.