Immune Modulation in Cancer
- Immunology, Infection and Cancer

Dr. Martina Seiffert
Head of Division
Immune surveillance plays a central role in controlling cancer initiation and progression, yet many tumors evade immune destruction by establishing immunosuppressive microenvironments. Our research aims to understand the cellular and molecular mechanisms underlying tumor-mediated immune escape, with a particular focus on T-cell exhaustion and the immunosuppressive functions of myeloid cells, in order to identify new strategies for improving cancer immunotherapies.
Image: Multiplex immunofluorescent image of a brain metastasis tissue section, © Liesa-Marie Pilger, in collaboration with Sascha Dietrich, Düsseldorf
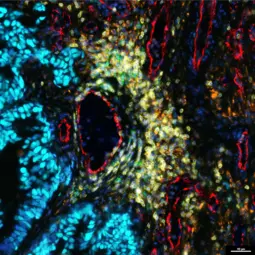
Image: Multiplex immunofluorescent image of a brain metastasis tissue section, © Liesa-Marie Pilger, in collaboration with Sascha Dietrich, Düsseldorf
The complex cellular and molecular composition of tumors is critical for their pathobiology. In previous studies, we have identified molecular mechanisms and candidate mediators responsible for cancer-induced remodeling of the tumor microenvironment and the immune system.
Our current research focuses on a detailed characterization of immune and stromal cells in B-cell lymphomas and brain metastases. The goal is to uncover novel pro-tumorigenic and immunosuppressive mechanisms, as well as to identify candidate genes and signaling pathways that may serve as targets for new therapeutic strategies. In addition, we investigate novel prognostic and predictive biomarkers and analyze mechanisms of therapy resistance.
A particular focus is on cancer-associated T-cell exhaustion, the immunosuppressive function of myeloid cells, and the mediators that control these phenotypes. Ongoing projects specifically address the roles of Interleukin-10 (IL-10), Interleukin-4-induced-1 (IL4I1), and Galectin-9.
To study these questions, we employ single-cell transcriptomics, spectral flow cytometry, and proteomic analyses of primary tumor tissues and blood cells. The spatial organization of the tumor microenvironment is analyzed using multiplex immunofluorescence staining and in situ transcript quantification in tissue sections. Functional analyses and treatment studies are conducted both in patient-derived in vitro systems and in mouse models to validate the relevance of identified candidate genes and pathways.
Our preclinical therapy studies also include optimization of treatment strategies for chronic lymphocytic leukemia (CLL) and diffuse large B-cell lymphoma (DLBCL) using CAR T-cell products and bispecific antibodies.
All of these efforts are carried out in close collaboration with academic and industrial partners and in continuous interaction with our clinical colleagues, to facilitate the rapid translation of our findings into novel therapeutic approaches.
Team
-

Dr. Martina Seiffert
Head of Division
-

Dr. Nicolas Aubert
-

Yuliia Borysovych
-

Hannah Briesch
-
Carolin Brombach
-

Covadonga Castellanos Gonzalez
-

Dr. Cèlia Dobano-López
-
Diego Duarte Gareta
-
Annika Fitz
-

Alessia Flörchinger
-
Sibylle Ohl
-

Liesa-Marie Pilger
-

Angelika Schermer
-
Johannes Schulze
Selected Publications
Llaó Cid L*, Wong JKL*, Fernandez Botana I, Paul Y, Wierz M, Pilger L-M, Flörchinger, A, Tan CL, Gonder, S, Pagano G, Chazotte M, Bestak K, Schifflers C, Iskar M, Roider T, Czernilofsky F, Bruch P-M, Mallm JP, Cosma, A, Campton DE, Gerhard-Hartmann E, Rosenwald A, Colomer D, Campo E, Schapiro D, Green EW, Dietrich S, Lichter P, Moussay E*, Paggetti J*, Zapatka M*, Seiffert M*
CD20-targeting bispecific antibodies improve response to CD19-CAR T-cells in-vitro and CLL in-vivo models
Brinkmann BJ*, Floerchinger A*, Schniederjohann C*, Roider T, Coelho M, Mack N, Bruch P-M, Liebers N, Dötsch S, Busch DH, Schmitt M, Neumann F, Roessner PM, Seiffert M*, Dietrich S*. *shared authorship
Interleukin-10 receptor signaling promotes the maintenance of a PD-1int TCF-1+ CD8+ T cell population that sustains anti-tumor immunity
Hanna BS*, Llao Cid L*, Iskar M, Roessner PM, Klett L, Wong JKL, Paul Y, Ioannou N, Öztürk S, Mack N, Kalter V, Colomer D, Campo E, Bloehdorn J, Stilgenbauer S, Dietrich S, Schmidt M, Gabriel R, Rippe K, Feuerer M, Ramsay A, Lichter P, Zapatka M, Seiffert M. *shared authorship.
IL4I1 is a metabolic immune checkpoint that activates the AHR and promotes tumor progression
Sadik A*, Somarribas Patterson LF*, Öztürk S*, Mohapatra SR*, Panitz V, Secker PF, Pfänder P, Loth S, Salem H, Prentzell MT, Berdel B, Iskar M, Faessler E, Reuter F, Kirst I, Kalter V, Foerster KI, Jäger E, Ramallo Guevara C, Sobeh M, Hielscher T, Poschet G, Reinhardt A, Hassel JC, Zapatka M, Hahn U, von Deimling A, Hopf C, Schlichting R, Escher BI, Burhenne J, Haefeli WE, Ishaque N, Böhme A, Schäuble S, Thedieck K, Trump S*, Seiffert M*, Opitz CA*. *shared authorship.
Get in touch with us

