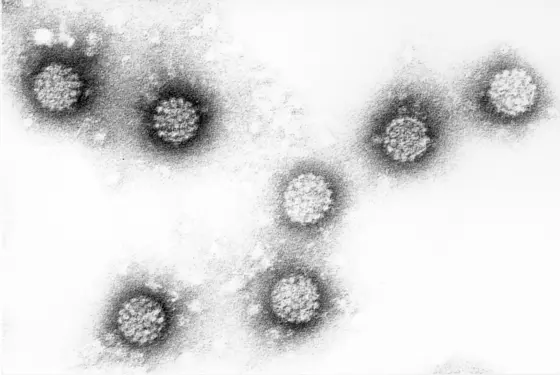
Oropharyngeal carcinoma, malignant tumors in the mouth and pharynx, are rare and are diagnosed in only 5 to 10 out of 100,000 inhabitants in Germany each year. Men are affected significantly more often than women. Alcohol and tobacco consumption as well as infection with carcinogenic human papillomaviruses (HPV) have been identified as risk factors for this cancer. In Germany, about half of all oropharyngeal cancers are due to HPV, mainly HPV16 is involved. However, the cancer has become more common in the Western world in recent years due to a rapid increase in the rate of HPV-related tumors.
Oropahrynx carcinomas do not form known precursor lesions and are therefore usually detected late, when they have already spread to adjacent lymph nodes. Scientists are therefore looking for biomarkers to identify high-risk patients for whom regular early screening by an otolaryngologist could help detect the dangerous disease as early as possible.
“Serum antibodies against the HPV16 protein E6 are detectable in more than 90 percent of all patients with HPV-related oropharyngeal cancer,“ says Tim Waterboer of the German Cancer Research Center. But the virologist and epidemiologist points to a fundamental problem with screening for rare diseases: “Because HPV-related oropharyngeal carcinomas are so rare in the general population, the positive predictive power of HPV16 E6 detection is comparatively low. That means, the vast majority people who have antibodies to HPV16 E6, will not develop HPV-related cancer.“
Waterboer an colleagues had made the observation that people with HPV-related oropharyngeal cancer not only form antibodies to HPV16 E6, but also to other HPV16 proteins formed during the early phase of viral infection (esp. E1, E2, E7). This situation is almost never found in individuals who do not have HPV-related tumors.
“Our hypothesis was that combined detection of antibodies to HPV16 E6 and at least one other early HPV16 protein could significantly improve the predictive power of a screening examination,“ Waterboer said.
Waterboer and colleagues were able to verify this assumption in participants of the Hamburg City Health Study, one of the largest health trials in Germany. Blood samples from 4424 participants recruited into the study between 2016 and 2017 were tested for the antibody combination. The researchers found antibodies to HPV16 E6 in 35 people (0.8 percent). But only eleven of the participants (0.3 percent) were seropositive for HPV16 E6 plus another early HPV16 protein.
These eleven high-risk individuals were subsequently invited to regular examinations at six-month intervals at the Department of Otolaryngology at the University Medical Center Hamburg-Eppendorf. Nine of the participants attended these examinations. During the following 1.3 years, HPV-related oropharyngeal carcinomas were diagnosed in three asymptomatic participants. The cancers were detected at stage 1 and were successfully treated.
“Early detection of these carcinomas significantly improves the quality of life of those affected. Typical consequences after the treatment of large tumors are, for example, swallowing and speech disorders, which massively reduce the quality of life of the patients. At an early stage, oropharyngeal carcinomas can be treated comparatively gently, so that patients have hardly any therapy-related restrictions,“ says Waterboer.
He emphasizes that the current work is a proof-of-concept study. “In many cases, there was an expectation that screening for oropharyngeal cancer would inevitably lead to an unacceptably high rate of false positive results due to the rarity of the disease. We were able to address that fear with the combined antibody analysis.“
Next, Waterboer and colleagues plan to include additional biomarkers and clinical examinations, as well as imaging techniques, to define the optimal conditions for screening for HPV-related oropharyngeal cancer. Only then will it be possible to assess whether a population-wide use of such a screening examination could be useful under medical and also economic aspects.
Eike Burandt, Krystle Lang Kuhs, Michael Pawlita, Tim Waterboer, Nicole Brenner: Detection of stage I HPV-driven oropharyngeal cancer in asymptomatic individuals in the Hamburg City Health Study using HPV16 E6 serology – a proof-of-concept study.
eClinical Medicine 2022, DOI: https://doi.org/10.1016/j.eclinm.2022.101659