Cellular immunotherapies that specifically target malignant tumors are thought to be a promising approach in cancer medicine. However, a basic requirement for this kind of targeted immunotherapy is to identify target molecules that are found exclusively on the tumor cells and are recognized by the immune system.
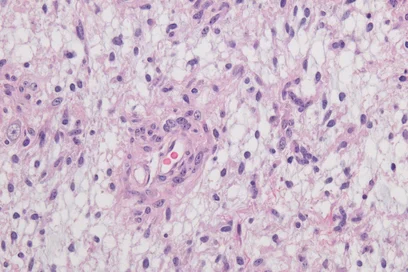
Malignant gliomas are incurable brain tumors that spread in the brain and cannot be completely removed by surgery. “Gliomas are very difficult to treat, and the lack of suitable target structures is a considerable challenge for developing immunotherapies,“ explained Lukas Bunse, an immunologist at the DKFZ and a doctor at University Hospital Mannheim (UMM).
Using an experimental mouse model, Bunse and his team have now demonstrated for the first time that transgenic T cells targeting tumor neoepitopes can be used to treat gliomas.
Tumor neoepitopes arise as a result of genetic mutations in cancer cells that lead to structural changes in the proteins produced. They therefore occur exclusively in the cancer cells. Using a prediction model, Bunse and his team identified a segment of the CIC protein (capicua transcriptional repressor) as a promising target structure for T cell attacks; around two percent of all gliomas show a recurrent mutation in this protein.
Mice vaccinated with the CIC neoepitope developed a population of T helper cells that showed a high level of activation in response to the vaccine peptide. The researchers used particularly active T cells to isolate the gene for the T cell receptor (TCR) responsible for epitope recognition.
They subsequently transferred the isolated TCR gene to cells and were thus able to grow large amounts of “transgenic“ T cells in a petri dish that all had an identical, highly active TCR targeting the CIC neoepitope.
In order to study their efficacy, the researchers injected the transgenic cells directly into the brain ventricles of glioma-bearing mice. In combination with radiotherapy, the T cell therapy led to glioma rejection in some of the animals.
“Here we have shown for the first time in an experimental model that a neoantigen-specific TCR-transgenic cell therapy can be effective against gliomas,“ explained Michael Kilian, first author of the study. “These kinds of neoepitope-specific TCR-transgenic T cells could be used in future in cancer patients who cannot be treated using CAR T cells,“ he added.
Chimeric antigen receptor (CAR) T cells, which have already been approved for treating patients with B cell leukemia, can only attack tumor antigens present on the surface of the cancer cells. However, these proteins are not usually found exclusively on tumor cells, so CAR T cells could damage healthy tissue too.
In contrast, the TCR-transgenic T cells can also attack mutated proteins from the cell interior, which must be exposed on the cell surface by special presentation molecules known as major histocompatibility complex (MHC) proteins. Most T cells respond solely to antigens presented by MHC molecules. To achieve results that can be transferred to humans, Lukas Bunse and his team therefore needed to work with mice transgenic for human MHC molecules.
“Our work suggests that TCR-transgenic T cells can also be used to treat patients with brain tumors,“ remarked Michael Platten, head of the Clinical Cooperation Unit Neuroimmunology and Brain Tumor Immunology at the DKFZ and Medical Director of Mannheim University Hospital's Department of Neurology. In collaboration with Wolfgang Wick, Medical Director of the Medical Faculty of Heidelberg's Department of Neurology, Platten and the team of neurooncologists hope to use comparable approaches to refine the TCR-transgenic T cell therapy and to study it in early clinical trials.
Michael Kilian, Mirco Friedrich, Khwab Sanghvi, Edward Green, Stefan Pusch, Daisuke Kawauchi, Martin Löwer, Jana K Sonner, Christopher Krämer, Julia Zaman, Stefanie Jung, Michael O Breckwoldt, Gerald Willimksy, Stefan B Eichmüller, Andreas von Deimling, Wolfgang Wick, Felix Sahm, Michael Platten, Lukas Bunse: T cell receptor therapy targeting mutant capicua transcriptional repressor in experimental gliomas
Clinical Cancer Research 2021, DOI: http://clincancerres.aacrjournals.org/content/early/2021/11/01/1078-0432.CCR-21-1881