Clue from laboratory mice on “junk food“
In order to investigate the immune system in NASH, the researchers fed laboratory mice a diet lacking essential nutrients but enriched with lipids and cholesterol – comparable to our “junk food“ – and observed the development of NASH. They studied the liver immune cells using single-cell RNA sequencing and discovered an unusually high number of a particular kind of cell, known as type 1 dendritic cells (cDC1), in the liver of NASH mice.
This phenomenon was not limited to mice. In tissue samples taken from patients in liver biopsies, the researchers found a correlation between the number of cDC1 cells and the extent of liver damage typical of NASH.
Do the cDC1 cells actually have an effect on liver pathology? The researchers pursued two channels of investigation here. They studied genetically modified mice lacking cDC1. In addition, they blocked cDC1 in the liver using specific antibodies. In both approaches, lower cDC1 activity was associated with a decrease in liver damage.
Dendritic cells normally only survive for a few days and need to be continually replaced by the immune system. The researchers discovered that the NASH-related tissue damage modulates the hematopoietic system in the bone marrow, as a result of which the cDC1 precursors divide more often and replenish the supply more readily.
Dendritic cells induce aggressive behavior in T cells
In a normal immune response, dendritic cells screen the organs for conspicuous immunologic features and then continue on to the neighboring lymph nodes – the command centers of the immune response – to pass on this information to the T cells. In NASH subjects, the German-Israeli team has now discovered that the cDC1 induce inflammatory and more aggressive behavior in T cells in the lymph nodes responsible for the liver, causing liver damage and leading to progression of the disease. “It is only recently that we identified these autoaggressive T cells as being responsible for liver damage in NASH. Now we also understand what induces this harmful behavior,“ Mathias Heikenwälder remarked.
Now that the cDC1 have been shown to play a key role in the progression of NASH, targeted manipulation of these cells might offer a new way of treating inflammation of the liver and its serious repercussions. “We are increasingly recognizing that certain cells of the immune system are involved in the development of different diseases, including cancer, diabetes, and Alzheimer's disease. Medicine is thus increasingly using ways of modulating the immune system and using drugs to push it in the right direction. This kind of approach might also work to prevent serious liver damage in NASH patients,“ Heikenwälder explained.
Eran Elinav, also a senior author of the study and head of research groups at DKFZ and the Weizmann Institute, believes that it is highly probable that gut bacteria affect the immune cells in this disease: “We now aim to find out how the gut and its bacteria influence activation of the immune cells in the liver. By doing so, we hope to be able to develop new treatment strategies.“
Why animal experiments are vital in cancer research
Liver cancer is a primary example of a type of cancer induced by chronic inflammation. Up until a few years ago, epidemiological studies showed chronic infection with hepatitis viruses to be the main driver of the disease. In recent years, however, the proportion of tumors linked to metabolism has steadily increased. Researchers are thus urgently looking for ways to understand more about this disastrous cascade of obesity-related cancer development in order to be able to provide treatments or – even better – to prevent it. To do so, they need to find out how obesity, which has a systemic impact on the entire organism, interferes with metabolic processes and immune reactions. These complex processes cannot be simulated in a petri dish; instead, the entire organism needs to be observed. Studies on mice are particularly well suited, as it is comparatively easy to genetically modify individual molecular drivers of the inflammatory process and hence identify the precise role they play.
Aleksandra Deczkowska, Eyal David, Pierluigi Ramadori, Dominik Pfister, Michal Safran, Baoguo Li, Amir Giladi, Diego Adhemar Jaitin, Oren Barboy, Merav Cohen, Ido Yofe, Chamutal Gur, Shir Shlomi-Loubato, Sandrine Henri, Yousuf Suhail, Mengjie Qiu, Shing Kam, Hila Hermon, Eylon Lahat, Gil Ben-Yakov, Oranit Cohen-Ezra, Yana Davidov, Mariya Likhter, David Goitein, Susanne Roth, Achim Weber, Bernard Malissen, Assaf Weiner, Ziv Ben-Ari, Mathias Heikenwälder*, Eran Elinav*, Ido Amit*: XCR1+ type 1 conventional dendritic cells drive liver pathology in non-alcoholic steatohepatitis
Nature Medicine 2021, DOI: https://www.nature.com/articles/s41591-021-01344-3
A picture is available for download:
NASH-cDCs-40X.jpg
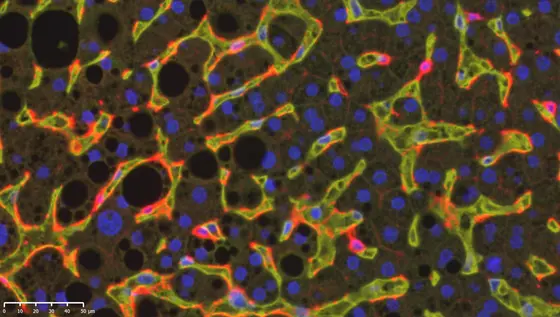
Caption: Immunofluorescence analysis of a NASH-affected human liver. DC1 cells (stained red, CD 141) patrol through the liver sinusoids (yellow, CD31). Black holes indicate lipid droplets.
Note on use of images related to press releases
Use is free of charge. The German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) permits one-time use in the context of reporting about the topic covered in the press release. Images have to be cited as follows: “Source: Heikenwälder / DKFZ“.
Distribution of images to third parties is not permitted unless prior consent has been obtained from DKFZ's Press Office (phone: ++49-(0)6221 42 2854, E-mail: presse@dkfz.de). Any commercial use is prohibited.