Genetic alterations often lead to cells producing altered proteins. This is particularly common in the so-called microsatellite unstable cancers: In these tumors, an important repair system has failed, which normally corrects small errors in the genetic material.If such DNA defects remain unrepaired, an additional nucleotide is often inserted into the DNA - with the result that the entire protein blueprint is altered.
The resulting novel protein structures, so-called neoantigens, are often recognized by the immune system as foreign. “It is well known that tumors with DNA repair defects, which have many neoantigens, also respond particularly well to immune therapies,“ explains the head of the study Matthias Kloor from Heidelberg University Hospital and the German Cancer Research Center.
About 15 percent of all cases of colorectal cancer and up to 30 percent of all uterine cancer are microsatellite unstable tumors. Until now, it was not known whether these neoantigens occur randomly in microsatellite unstable cancers.
Kloor and his team now systematically analyzed 139 microsatellite unstable tumors. To do this, the researchers used an algorithm newly developed at the University Hospital, the DKFZ, the Interdisciplinary Center for Scientific Computing at the University of Heidelberg and the HITS, which quantitatively evaluates the mutations in tumor cells. In a second step, the scientists were able to predict which of these neoantigens are capable of activating the immune system.
The surprising result of the investigation: The mutations do not occur randomly at any point in the genome. Rather, the scientists found numerous specific recurring genetic mutations that were identical in the tumors of many patients. Many of these matching mutations lead to the formation of neoantigens which, according to bioinformatic predictions, are able to activate the immune system and thus mobilize against the cancer.
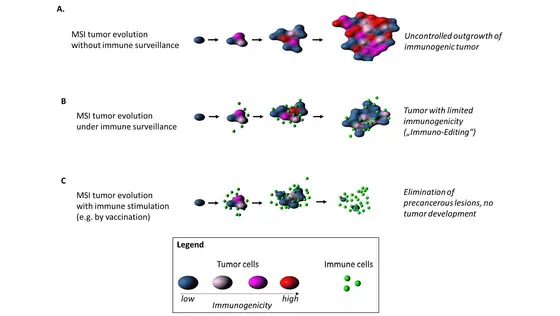
The second surprise: Mutations that lead to strongly immunogenic neoantigens are actually rather rare in microsatellite unstable (MSI) cancers. “This may indicate that the immune system monitors the tumor during its development and immediately eliminates cancer cells with highly immunogenic neoantigens. The tumor then consists mainly of cells whose neoantigens activate the immune system much less. The immune system therefore forms the tumor during its development,“ explains Matthias Kloor. However, there are certain mutations that are frequently found in MSI tumors despite the high immunogenicity of the resulting neoantigens. These mutations seem to drive tumor development. Such neoantigens resulting from mutations with tumor-driving effect are particularly promising for vaccine development.
“This observation confirms our idea that it might be possible to prevent the development of clinically relevant tumors with vaccinations against selected neoantigens,“ says Magnus von Knebel Doeberitz, also author of the paper and head of a research department located both at the DKFZ and at Heidelberg University Hospital.
Patients with Lynch's syndrome, in whom DNA repair defects occur within the family and who therefore often develop cancer at a younger age, could particularly benefit from this approach. The researchers' idea is to sensitize the immune system of these patients specifically to those neoantigens that result from mutations that particularly drive malignant cancer growth. This could influence tumor development in such a way that the outgrowth of dangerous cancer cell clones becomes much less likely. Before clinical application, however, it is necessary to test this preventive approach for its efficacy in further pre-clinical and clinical studies.
Original publication:
Alexej Ballhausen, Moritz Jakob Przybilla, Michael Jendrusch, Saskia Haupt, Elisabeth Pfaffendorf, Markus Draxlbauer, Florian Seidler, Sonja Krausert, Aysel Ahadova, Martin Simon Kalteis, Daniel Heid, Damian Stichel, Johannes Gebert, Maria Bonsack, Sarah Schott, Hendrik Bläker, Toni Seppälä, Jukka-Pekka Mecklin, Sanne Ten Broeke, Maartje Nielsen, Vincent Heuveline, Julia Krzykalla, Axel Benner, Angelika Beate Riemer, Magnus von Knebel Doeberitz, Matthias Kloor: The shared neoantigen landscape of MSI cancers suggests immunoediting during tumor evolution
Nature Communications 2020, DOI: 10.1038/s41467-020-18514-5.