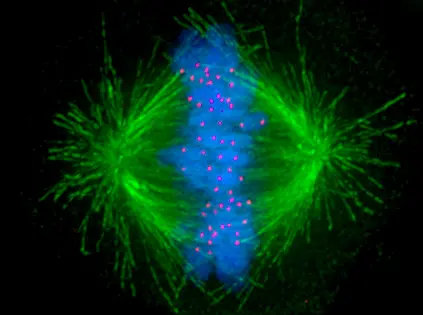
Before cells divide, they must first ensure that their genetic material is correctly distributed to both daughter cells. To achieve this, they build up a bundle of special protein microtubules known as mitotic spindle because of its characteristic shape. The microtubules of the spindle pull the individual chromosomes in an ordered fashion to the opposite poles of the cell and are then normally degraded.
Many classical anti-cancer drugs act on the spindle microtubules: The taxanes originally obtained from the yew, for example, prevent the degradation of the microtubules and thus stabilize the mitotic spindle. As a result, the cancer cells remain stuck in cell division. Normally, this triggers cell death.
“But after prolonged administration of these drugs, cancer cells can escape the mitosis stop, return to the cell cycle and continue to divide - and thus develop resistance to chemotherapy with taxanes,“ explains Ingrid Hoffmann from the German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ).
In her current work on cancer cell lines, the scientist and her team therefore investigated the protein FBXW7, which is responsible for mitosis stop-induced cell death. FBXW7 is therefore considered a tumor suppressor gene. The researchers found that the longer the mitosis stop lasts, the more FBXW7 concentration in the cancer cells decreases.
On closer examination, the scientists found that the FBXO45-MYCBP2 protein complex attaches to FBXW7 during the mitosis stop. This complex is part of the cell's recycling system: it attaches the signal molecule ubiquitin to other proteins and labels them for degradation. This reduces the amount of FBXW7 available and the cancer cells escape cell death.
Using micro-RNAs, the researchers were able to genetically block the FBXO45-MYCBP2 complex and thus stop the degradation of the tumor suppressor. This leads to increased tumor cell death and prevents the development of resistance.
“This is the first time that we have described a surprising connection between protein degradation and the development of resistances against taxanes and other drugs that act on the spindle microtubules,“ said Hoffmann. “Our result may possibly contribute to the development of more effective drug treatments in the future that circumvent this form of resistance.“
Kai T. Richter, Yvonne T. Kschonsak, Barbara Vodicska and Ingrid Hoffmann: FBXO45-MYCBP2 regulates mitotic cell fate by targeting FBXW7 for degradation.
Cell Death and Differentiation 2019, DOI: 10.1038/s41418-019-0385-7