In juvenile myelomonocytic leukemia (JMML), blood cells grow out of control. This rare type of blood cancer affects primarily children, mostly aged two and younger. Without stem cell transplantation (also called bone marrow transplant), which is currently the only treatment option that has been effective against the cancer, the disease takes a fatal course in most cases.
In about one third of the patients who have received a transplant, the cancer relapses. Therefore, it is crucial to make exact predictions about the course of the disease early on. The most recent findings by researchers in the team of Christoph Plass from the German Cancer Research Center (DKFZ) in Heidelberg and by a team led by Christian Flotho, scientists from the German Cancer Consortium (DKTK) in Freiburg, may now help better assess the course of JMML cases in the future. “Clinical parameters have only facilitated inaccurate predictions so far,“ said Plass. “Comprehensive molecular analyses like ours may help predict more accurately whether the cancer responds to therapy.“
The investigators examined the complete genome of JMML patients for specific tags called DNA methylations. DNA methylation, as well as other chemical modifications of the genome, are called epigenetic changes. They regulate the activity of individual genes. In contrast to genetic mutations, the DNA sequence remains unchanged in epigenetic regulation.
In their study, the scientists discovered that JMML patients can be classified in three subgroups, depending on their DNA methylation levels. “Until now, only specific genes, and not the whole genome, have been examined for methylations in JMML,“ Plass said.
The researchers found out that patients whose genomes exhibit high levels of methylation also tend to show characteristics that are linked to an increased risk of relapsing after bone marrow transplant. By contrast, there is a patient group whose DNA exhibits only a few methylations and whose disease takes a milder course. A third group exhibits medium levels of methylation. “These patients benefit from stem cell transplantation and usually have a more favorable prognosis with this treatment,“ said Daniel Lipka, who is the first author of the study.
“Analyzing the DNA methylation status might help better predict the course that the disease will take in an individual patient,“ Lipka said. “We now plan to conduct further studies with larger patient numbers in order to characterize the three groups more precisely. This might make it possible in the future to adjust therapies early on.“
Daniel B. Lipka, Tania Witte, Reka Toth, Jing Yang, Manuel Wiesenfarth, Peter Nöllke, Alexandra Fischer, David Brocks, Zuguang Gu, Jeongbin Park, Brigitte Strahm, Marcin Wlodarski, Ayami Yoshimi, Rainer Claus, Michael Lübbert, Hauke Busch, Melanie Boerries, Mark Hartmann, Maximilian Schönung, Umut Kilik, Jens Langstein, Justyna A. Wierzbinska, Caroline Pabst, Swati Garg, Albert Catalá, Barbara De Moerloose, Michael Dworzak, Henrik Hasle, Franco Locatelli, Riccardo Masetti, Markus Schmugge, Owen Smith, Jan Stary, Marek Ussowicz, Marry M. van den Heuvel-Eibrink, Yassen Assenov, Matthias Schlesner, Charlotte Niemeyer, Christian Flotho, Christoph Plass: RAS-pathway mutation patterns define epigenetic subclasses in juvenile myelomonocytic leukemia
Nature Communications 2017, DOI 10.1038/s41467-017-02177-w
An image for this press release is available for download at:
JMML-16-PB.jpg
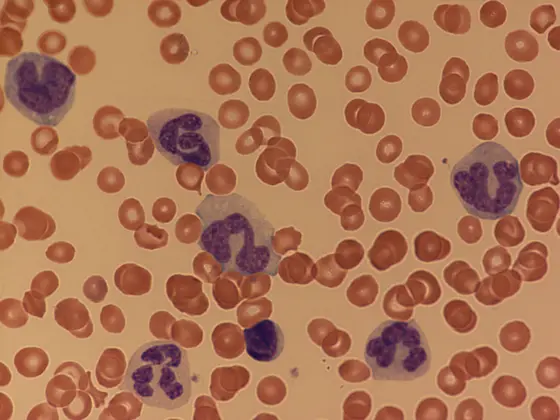
Caption: The peripheral blood of JMML patients contains far too many immature myeloid and monocytic cells.
Note on use of images related to press releases
Use is free of charge. The German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) permits one-time use in the context of reporting about the topic covered in the press release. Images have to be cited as follows: “Source: Universitätsklinikum Freiburg“.
Distribution of images to third parties is not permitted unless prior consent has been obtained from DKFZ's Press Office (phone: ++49-(0)6221 42 2854, E-mail: presse@dkfz.de). Any commercial use is prohibited.