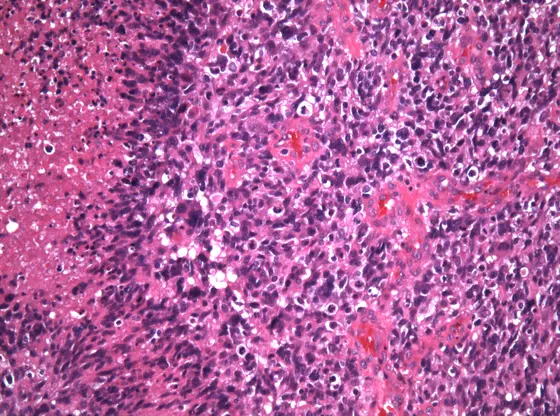
Glioblastoma is one of the most dangerous and aggressive types of brain cancer. Although it occurs mostly in adults, each year approximately 60 children and adolescents in Germany are also diagnosed with this dreaded form of cancer. Established radiotherapy and chemotherapy treatments can usually only slightly delay the progression of the disease. Scientists are therefore urgently searching the genomes of glioblastoma cells for cancer-promoting alterations that can be attacked with modern targeted drugs.
David Jones and his colleagues at the German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) in Heidelberg analyzed the genomes in more than 50 cases of pediatric glioblastoma, in a study published in Nature Medicine. The study was conducted within the German PedBrain Tumor Research Project, which is part of the International Cancer Genome Consortium (ICGC) and focuses on pediatric brain tumors. The researchers identified a large variety of defects that cause the cells to turn cancerous. In ten percent of the cases, which is a remarkable rate, they discovered a defect where a known cancer gene called MET was fused with other DNA segments.
“Fusions of growth-promoting genes are typical for cancer,“ said Marie-Laure Yaspo from the Max Planck Institute for Molecular Genetics (MPIMG) in Berlin, whose group confirmed the gene fusion discovery. “The most well-known is the so-called 'Philadelphia' chromosome, or BCR-ABL fusion, which is the cause of almost all cases of chronic myeloid leukemia.“ These gene fusions lead to cancer when a growth-promoting gene that is normally under strict control fuses with other genes that lead to abnormal activation.
MET genes that have undergone mutations or amplification have been described as tumor promoters in other cancer types. MET is one of the receptor proteins in the cell membrane that respond to growth factors. Many of these receptors, including MET, can now be blocked using inhibitors that have been approved as drugs. “In order to be able to treat affected children with these substances, we first needed to prove that MET fusion genes in fact promote the development of glioblastoma,“ Jones said.
To provide this proof, the investigators transferred MET fusion genes to the respective brain cells in mice. The transferred genes subsequently caused exactly this type of brain cancer – glioblastoma. In further experiments with these animals, the researchers were able to confirm that MET inhibitors significantly slowed down cancer growth.
Children with relapsing cancer mostly have a very small chance of being permanently cured. To improve the curative prospects for these young patients, Heidelberg pediatric oncologists and molecular biologists are coordinating a registry study called INFORM that pursues molecular analysis of tumor genomes. The INFORM project is being carried out within the framework of the German Cancer Consortium (DKTK). In an eight year-old INFORM patient who suffered from glioblastoma, the genome analysis had detected a MET fusion gene. As part of a personalized therapy approach, the physicians treated the child with a MET inhibitor called crizotinib. As a result, the tumor shrunk substantially and the severe disease symptoms improved. At the same time, however, further tumors rapidly formed in the patient's brain which failed to respond to the treatment.
“The drug was effective, but unfortunately it was insufficient,“ said Stefan Pfister, a pediatric oncologist and molecular biologist at the DKFZ and Heidelberg University Hospital. “Resistance against MET inhibitors is a known problem. It is becoming increasingly clear to us that we have to combine these drugs with other agents.“ In addition, the MET fusion genes were not the only molecular culprits driving cancer growth. In all tumors, the researchers had also found mutations in genes that control the cell cycle. “It would surely be useful to combine MET inhibitors with agents that normalize the deregulated cell cycle,“ Pfister said.
The PedBrainTumor Project has been supported by the Federal Ministry of Education and Research (BMBF) and German Cancer Aid (Deutsche Krebshilfe).
Sebastian Bender, Jan Gronych, Hans-Jörg Warnatz, Barbara Hutter, Susanne Gröbner, Marina Ryzhova, Elke Pfaff, Volker Hovestadt, Florian Weinberg, Sebastian Halbach, Marcel Kool, Paul A. Northcott, Dominik Sturm, Lynn Bjerke, Thomas Zichner, Adrian M. Stütz, Kathrin Schramm, Bingding Huang, Ivo Buchhalter, Michael Heinold, Thomas Risch, Barbara Worst, Cornelis M. van Tilburg, Ursula D. Weber, Marc Zapatka, Benjamin Raeder, David Milford, Sabine Heiland, Christof von Kalle, Christopher Previti, Chris Lawerenz, Andreas E. Kulozik, Andreas Unterberg, Olaf Witt, Andreas von Deimling, David Capper, Nathalène Truffaux, Jacques Grill, Nada Jabado, Astrid M. Sehested, David Sumerauer, Dorra Hmida-Ben Brahim, Saoussen Trabelsi, Ho-Keung Ng, David Zagzag, Jeffrey Allen, Matthias A. Karajannis, Nick Gottardo, Chris Jones, Jan O. Korbel, Sabine Schmidt, Stephan Wolf, Guido Reifenberger, Jörg Felsberg, Benedikt Brors, Christel Herold-Mende, Hans Lehrach, Tilman Brummer, Andrey Korshunov, Roland Eils, Marie-Laure Yaspo, Stefan M. Pfister, Peter Lichter, and David T.W. Jones for the International Cancer Genome Consortium PedBrain Tumor Project36: Recurrent MET fusion genes represent a druggable target in paediatric glioblastoma.
Nature Medicine 2016, DOI: 10.1038/nm.4204.
About the INFORM registry:
http://www.dkfz.de/de/inform/ueber-inform.html