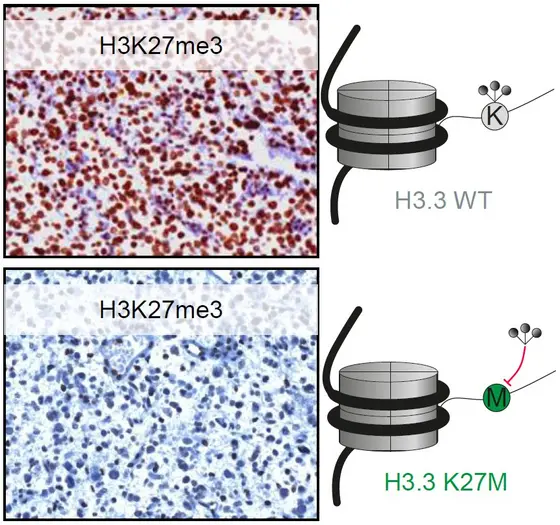
Highly malignant glioma is one of the most dangerous types of pediatric brain cancer. Less than ten percent of children and adolescents who suffer from this disease survive the first three years after diagnosis. Systematic analyses of the DNA of this type of tumor have revealed that approximately 50 percent of the cases are characterized by a mutation in a gene encoding one of the proteins responsible for DNA packaging, a histone called H3.3. In the amino acid chain of the mutated protein, the amino acid lysine (K) has been replaced by a methyonine (M) in position 27 (and thus is called the “K27M mutation").
This replacement has an impact not only on the mutated packaging protein itself, but also on other H3 histone proteins. In tumor cells with the K27M, the mutation leads to significantly reduced numbers of methyl groups on H3.3 and other histone proteins as well.
Professor Stefan Pfister of the DKFZ coordinated an international project with the goal of analyzing how the K27M mutation affected malignant gliomas. First, the investigators used a special immune test to study the amount of methylation on histone proteins. They examined samples from more than one hundred malignant gliomas which had been analyzed to determine whether they carried mutations in H3.3. The analysis showed a nearly 100 percent correlation between K27M mutations in gliomas and low levels of H3 histone methylation.
In the current article, the researchers also shed light on the mechanisms underlying these variations in histone methylation: The H3.3 histone mutant binds to an enzyme called EZH2 at higher than normal levels; EZH2 is a methyl transferase, which is responsible for transferring methyl groups onto other molecules. The binding of the two molecules subsequently reduces the enzymatic activity of EZH2.
Methyl groups can be attached to DNA as well as proteins, and the K27M mutation affects the methylation of both types of molecules in gliomas that carry it. Levels of methylation are lower than in tumor cells without the mutation. Both of these epigenetic phenomena lead to an increase in gene activity in the malignant brain tumors, which, in turn, may have an impact on the aggressiveness of the cancer.
“Variations in H3 histone methylation patterns have also been described in other types of cancer, where they are often caused by defects in the gene coding for the EZH2 methyltransferase itself," says Sebastian Bender, first author of the article. “Our article connects a specific genetic change that is found in aggressive gliomas to its impact on epigenetic mechanisms such as histone and DNA methylation."
Sebastian Bender et al.: Reduced H3K27me3 and DNA Hypomethylation Are Major Drivers of Gene Expression in K27M-Mutant Pediatric High-Grade Gliomas. Cancer Cell 2013, DOI: 10.1016/j.ccr.2013.10.006