The immune system usually recognizes foreign substances, such as pathogens and destroys them. It can also recognize cancer cells, even though they arise from the body, because their DNA and protein composition have undergone extreme transformations. This is accomplished by immune cells called “killer T cells" that invade the tumor tissue. If this doesn’t happen naturally, there are ways to boost their activity; several years ago, researchers succeeded in “training" killer T cells to attack tumor tissue in the test tube. But when such activated cells are injected into patients, they are often ineffective, because many of them fail to reach the tumor.
Normally, immune cells migrate into tissue through “anchors" formed by blood vessels. As the cells flow by, they latch onto the anchors and can thus leave the bloodstream. However, tumors often prevent the anchors from forming, which prevents the killer T cells from using them as exit points. The Heidelberg researchers, headed by Professor Philipp Beckhove, found a way to intervene. Their idea was to focus low doses of radiation at specific points in a tumor, provoking an immune response that would lead to the formation of anchor molecules in the vessel walls. “This strategy has been successful in mice with pancreatic cancer," says Beckhove. “Anchor molecules started forming again in irradiated tumors. In addition, the tumors accumulated four times as many killer T cells than those that had not been irradiated." When the scientists additionally administered activated killer T cells, these, too, invaded the tumor. What is more: “The mice survived at least ten weeks longer than animals treated exclusively with either radiation therapy or activated T cells," Beckhove says.
Beckhove and his team also thought that macrophages, another type of immune cell, might be involved in this radiation-induced immune response. “It was known that macrophages accumulate in tumor tissue," says Beckhove. “But they have been thought to do more harm than good." To unravel the role of macrophages, the investigators conducted another experiment where they irradiated macrophages from healthy mice. These were subsequently injected into cancerous mice along with activated T cells. The result: once again, the killer T cells migrated into the tumor tissue and attacked the tumor. The researchers thus proved that changes in macrophages, caused by irradiation, trigger the immune response. “Amazingly, a single gene called iNOS is responsible for all this," Beckhove says. “It is activated in macrophages when they are exposed to radiation."
The first clinical trials with patients suffering from pancreatic or bowel cancer demonstrate that irradiation also attracts killer T cells to tumors. Prior to surgery, scientists treated patients with a dose of radiation 10- to 30-times lower than normal and examined the tumor after resection. “We hope that this low-dose irradiation also has a positive effect on the course of the disease in human cancers," says Beckhove. “If so, it might be used to treat inoperable tumors, in order to increase the effectiveness of subsequent immunotherapy."
Felix Klug, Hridayesh Prakash, Peter E. Huber, Tobias Seibel, Noemi Bender, Niels Halama, Christina Pfirschke, Ralf Holger Voss, Carmen Timke, Ludmila Umansky, Kay Klapproth, Knut Schäkel, Natalio Garbi, Dirk Jäger, Jürgen Weitz, Hubertus Schmitz-Winnenthal, Günter J. Hämmerling and Philipp Beckhove: Low-Dose Irradiation Programs Macrophage Differentiation to an iNOS+/M1 Phenotype that Orchestrates Effective T Cell Immunotherapy, Cancer Cell, DOI: 10.1016/j.ccr.2013.09.014.
A picture for this press release is available at:
immuntherapie-blutgefaesse.jpg
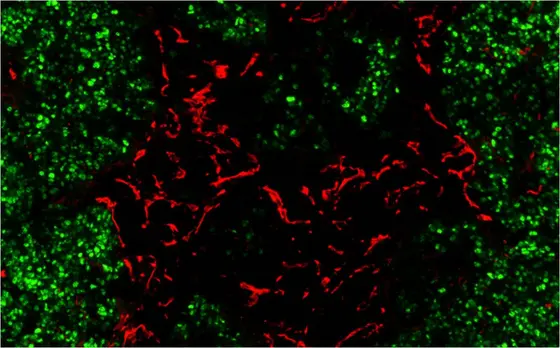
Red: A big problem for immunotherapy of cancer are blood vessels that are not properly formed. Green: Tumor cells