White adipose tissue is the body’s energy reservoir. It is where excess fat is stored for use in times of hunger to supply other organs, particularly skeletal muscle, with energy. After meal, insulin causes fat molecules from the blood to be stored in the adipose cells. When fasting, on the other hand, adrenalin and noradrenalin cause fat to be broken down into glycerol and fatty acids. If this balance between storage and decomposition is disrupted, obesity and adverse health effects associated with it may result: The adipose tissue produces more hormones which further promote overweight, inflammatory parameters increase and the regulation of blood glucose levels gets out of control.
“These are the typical first symptoms of type 2 diabetes," says Prof. Dr. Stephan Herzig, a metabolism expert who heads a joint research department of the German Cancer Research Center (DKFZ), Heidelberg University and Heidelberg University Hospital. He is looking for molecular switches that control metabolism. TBLR1 is one of these. Herzig and his team have recently shown that mice with genetically silenced TBLR1 develop fatty liver. The Heidelberg researchers have now studied whether TBLR1 starts the biochemical signaling cascades in adipose cells ultimately leading to activation of lipases, fat-breaking-down enzymes.
The researchers bred mice whose fat cells couldn´t produce TBLR1. These animals do not break down their fat deposits even after 24 hours of fasting. When fed a fat-rich diet, TBLR1-deficient mice gained much more weight than healthy control animals. At the same time, their glucose metabolism was disrupted and the adipose tissue contained more proinflammatory molecules. “Lipid metabolism as a whole gets into a critical, pathogenic state," says Stephan Herzig summing up these molecular changes.
Herzig’s team went on to investigate whether TBLR1 controls fat decomposition in humans, too. The researchers analyzed adipose tissue of women who kept a strict diet for weight loss. At the end of this diet, their adipose cells contained much more TBLR1 RNA than before. Hunger apparently stimulates production of this gene switch, which, in turn, stimulates fat decomposition, also in humans. At the same time, levels of inflammatory marker CRP decreased and the adipose cells produced more hormones with health-promoting effect.
“TBLR1 appears to counteract harmful metabolic changes which are typical for overweight people and can lead to serious diseases," says Stephan Herzig. A long-term goal of his research is to use molecular switches such as TBLR1 to specifically influence a disrupted energy metabolism.
Maria Rohm, Anke Sommerfeld, Daniela Strzoda, Allan Jones, Tjeerd Sijmonsma, Gottfried Rudofsky, Christian Wolfrum, Carsten Sticht, Norbert Gretz, Maximilian Zeyda, Lukas Leitner, Peter Nawroth, Thomas Stulnig, Mauricio Berriel Diaz, Alexandros Vegiopoulos and Stephan Herzig: Transcriptional co-factor TBLR1 controls lipid mobilization in white adipose tissue. Cell Metabolism 2013,doi: 10.1016/j.cmet.2013.02.010.
A picture is available at the internet:
WAT-lean-obese.jpg
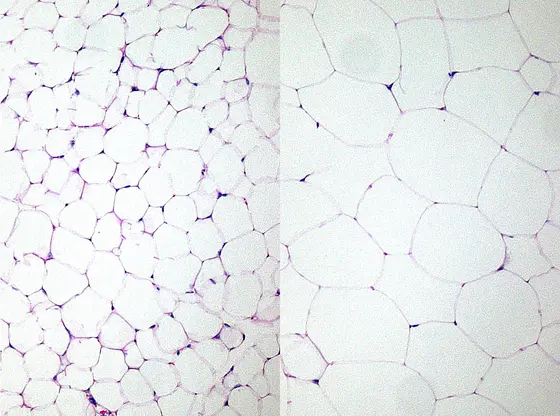
Picture caption: White adipose tissue of a lean (left) and an obese mouse
Picture Source: Maria Rohm, Deutsches Krebsforschungszentrum