Poor blood circulation and increased metabolic activity often create hostile conditions in tumors: typical symptoms include a lack of oxygen, glucose, and other nutrients, the accumulation of sometimes harmful metabolites, and acidification of the tumor environment. Experts refer to this as acidosis.
The team led by Wilhelm Palm from the DKFZ and Johannes Zuber from the IMP investigated how cancer cells adapt to these harsh conditions. First, the researchers systematically switched off each gene individually in pancreatic cancer cells using the CRISPR-Cas9 gene editing tool and then tracked how its loss affected the survival and growth of the cells under defined stress conditions. These experiments were initially conducted in culture dishes. The genes identified using this approach were then specifically switched off in mice with pancreatic cancer, and the effects were compared with the results from the cell culture.
The comparative analysis of hundreds of such genes relevant to cancer cell growth under stress conditions surprisingly showed that the metabolism of cancer cells in the mouse model was strongly influenced by adaptations of their energy balance to tumor acidosis. The metabolism of cancer cells within a tumor differs significantly from that in conventional cell culture and can best be replicated by an acidic environment.
“It is not just the lack of oxygen or nutrients that changes the metabolism in the tumor – it is primarily the acidification of the tumor environment,” explains Wilhelm Palm. Acidosis helps cancer cells switch from sugar-based energy production (glycolysis) to more efficient energy production through respiration in the mitochondria. These cell structures, known as organelles, are also referred to as the “powerhouses of the cell.”
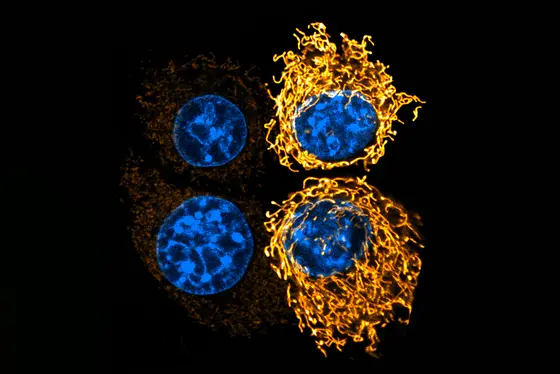
The researchers were able to show that the acidic pH value triggers profound changes in the mitochondria. Normally, they are present in cancer cells as small, fragmented structures. Under acidic conditions, however, they merge into extensive networks that are significantly more efficient.
This is possible because acidosis inhibits the activity of the signaling protein ERK. Overactivation of this signaling pathway normally causes mitochondria in cancer cells to repeatedly divide into many small fragments. If this fragmentation does not occur as a result of tumor acidosis, mitochondria can use various nutrients more efficiently for energy production. If genetic intervention prevents the mitochondria from fusing, cancer cells lose their metabolic flexibility and grow much more slowly in the acidic environment of a tumor.
“Our results show that acidosis is not simply a by-product of tumor metabolism, but an important switch that controls the energy supply and survival strategies of cancer cells,” explains co-study leader Johannes Zuber. In the long term, these findings could open up new avenues for therapies that specifically target the energy metabolism of tumors.
Publication:
Groessl S, Kalis R, Snaebjornsson MT, Wambach L, Haider J, Andersch F, Schulze A, Palm W, Zuber J. Acidosis orchestrates adaptations of energy metabolism in tumors. Science 2025, DOI: 10.1126/science.adp7603
A picture is available for download:
https://www.dkfz.de/fileadmin/user_upload/Skoe/Pressemitteilungen/2025/Picture_PR.jpg
Caption: Cancer cells reshape their mitochondria (stained yellow) when exposed to acidosis. The composed image shows two cells under neutral pH (left) compared to an acidic environment (right), where mitochondria form elongated networks.
Note on use of images related to press releases
Use is free of charge. The German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) permits one-time use in the context of reporting about the topic covered in the press release. Images have to be cited as follows: “Source: Groessl / DKFZ”.
Distribution of images to third parties is not permitted unless prior consent has been obtained from DKFZ’s Press Office (phone: ++49-(0)6221 42 2854, E-mail: presse@dkfz.de). Any commercial use is prohibited.