Neuroimmunologie und Hirntumorimmunologie
- Immunologie, Infektion und Krebs
- Klinische Kooperationseinheit

Prof. Dr. med. Michael Platten
Department Head
„Wir entwickeln innovative personalisierte Immuntherapien für Patient*innen mit Hirntumoren und überprüfen und verbessern diese in klinischen Studien."

Unsere Forschung
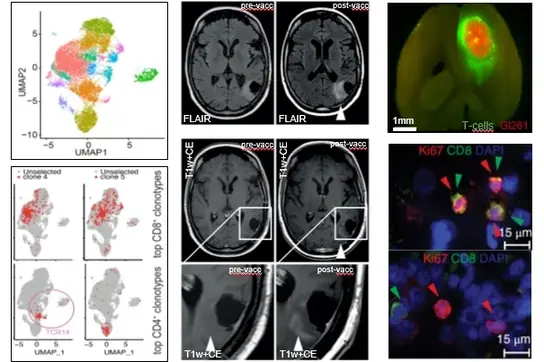
Das Zentralnervensystem (ZNS) wird als ein immunprivilegiertes Organ betrachtet, in dem Immunantworten trotz der Blut-Hirn-Schranke über einen intensiven Austausch mit dem peripheren Immunsystem streng kontrolliert werden. Unsere Gruppe entwickelt und verbessert immuntherapeutische Ansätze zur Bekämpfung von Hirntumoren mithilfe der Analyse molekularer Mechanismen der Immunsuppression und indem wir neuartige immuntherapeutische Behandlungsmodalitäten nutzen. Unsere Expertise liegt in umfassender zellulärer und auf Bildgebung basierender Analyse der Tumormikromilieus, Transcriptomics, sowie Profiling und Nutzung von Immunrezeptoren, sowohl in Mausmodellen und anhand von klinischen Proben. Unser zentraler Fokus ist die klinische Translation.
In den letzten Jahren haben wir immunsuppressive Funktionen und Mechanismen zweier von Hirntumoren produzierter Schlüssel-Metabolite identifiziert. Unsere Entdeckung, dass aus TDO entstandeneTryptophanmetabolite (Kynurenine) Tumorwachstum und Immunsuppression durch die Aktivierung des Arylhydrocarbonrezeptors (AhR) vermitteln, ermöglichte die Nutzung neuer therapeutischer Zielstrukturen und implizierte weitere Fragen, die wir aktuell in Tumormodellen adressieren. Ein zentrales Ziel ist die Identifizierung von Substanzen, die in den Tryptophankatabolismus eingreifen, um so potentielle Therapeutika für maligne Gliome zu entwickeln.
Weiterhin haben wir gezeigt, dass 2-Hydroxyglutarat, welches durch IDH-Mutationen von Gliomzellen produziert wird, direkt adaptive zelluläre Immunantworten, aber auch das inerte Immunsystem im Mikromilieu des Hirntumors inhibiert. Diese Erkenntnisse ebnen den Weg zu neuartigen Konzepten der immuntherapeutischen Kombinationstherapie, die wir momentan in präklinischen Gliommodellen untersuchen und in ihre klinische Translation begleiten.
Die Entwicklung neuartiger Zielantigene und T-Zellrezeptoren für die zielgerichtete Immuntherapie von Hirntumoren wurde in klinische Studien umgesetzt um die therapeutische Relevanz von mutationsspezifischen Vakzinen gegen klonale Hirntumor „Driver“ Mutationen zu demonstrieren. Laufende Projekte sind nun auf die Identifikation weiterer mutierter Antigene für die spezifische Immuntherapie und auf die Entdeckung spezifischer T-Zellrezeptoren (TZR) für die transgene T-Zelltherapie für Gliompatienten fokussiert. Unser Fokus liegt hierbei weiterhin bei CD4+ T-Helferzellen, die lange Zeit bezüglich ihrer Rolle bei der Immuntherapie vernachlässigt wurden. Hierfür haben wir einen Arbeitsablauf zur Entwicklung einer patientenspezifischen zielgerichteten Immuntherapie für Patienten mit Gliomen auf der Basis von Mutanomanalysen und der Identifikation von TZR, sowie ein MHC-humanisiertes murines Gliommodell zur präklinischen Evaluation entwickelt.
Bioinformatische Selektion reaktiver, in den Tumor infiltrierender T-Zellen zur TZR-Identifikation, basierend auf moderner Hochdurchsatz-Einzelzell-RNA-Sequenzierung, Einzelzell-VDJ-Sequenzierung sowie TZR-beta deep Sequenzierung, treiben diese Prozesse an und fördern ihre Entwicklung.
Unser Forschungsfokus
Wir entwickeln von der präklinischen Validierung in humanisierten Mausmodellen bis hin zu klinischen Studien lange Peptid Vakzine gegen die klonalen Driver Mutationen wie IDH1.R132H oder H3.K27M in Hirntumoren. Die klinischen Studien haben gezeigt, dass vakzininduzierte mutationsspezifische CD4 T-Zellen in der Lage sind, in das zentrale Nervensystem einzuwandern und eine therapeutische Entzündungsreaktion auszulösen. Die iterative Optimierung von Vakzinantworten ist uns besonders wichtig, daher kombinieren wir unsere Vakzine mit Immuncheckpoint Inhibitoren und testen diese in sogenannten “window-of-opportunity” Studien für die rückwärtige Translation. Hochdurch-Einzelzell-Sequenzierungen von peripheren und tumor-infiltrierenden T-Zellen erlauben uns dabei die Patienten individuelle Charakterisierung und Validierung von Hirntumor spezifischen T-Zellen, die für das Engineering von T-Zellen verwendet werden können.
Um immuntherapeutische Ansätze vom Labor ans Krankenbett zu übertragen, müssen wir die tumorspezifischen Immunantworten verstehen und das gesamte Wirksamkeitsspektrum von der Tumorregression bis zur Resistenz untersuchen. Dies wird durch viele patientenindividuelle Faktoren erschwert, so dass ein sorgfältiges Studiendesign und empfindliche Assays unerlässlich sind.
Wir sind Experten für die Überwachung des menschlichen Immunsystems und arbeiten eng mit unseren klinischen Partnern zusammen, um ein tiefes Profiling der individuellen Immunreaktionen zu erreichen. Wir haben eine Reihe sensitiver funktioneller Assays (ELISpot, ELISA, TIL-Expansion, epitop-spezifische Expansion, Durchflusszytometrie) entwickelt, die bei verschiedenen Arten von Patientenproben (Blut, Liquor, Tumorgewebe) zum Einsatz kommen.
Parallel dazu legen wir besonderen Wert auf hochmoderne Sequenzierungstechnologien, wie z. B. Massen- und Einzelzell-TCR- und RNA-Sequenzierung, um die Dynamik der T-Zellreaktionen zu verstehen. So können wir antigen- oder behandlungsbedingte Veränderungen in der Zusammensetzung des Repertoires beobachten und einzelne T-Zell-Klonotypen über verschiedene Proben/Gewebe und Zeitpunkte hinweg verfolgen und charakterisieren. TCR-Kandidaten mit mutmaßlicher Anti-Tumor-Aktivität können identifiziert, rekombinant exprimiert und in vitro im Hinblick auf Spezifität, Affinität, Avidität und HLA-Restriktion charakterisiert werden. Wir verwenden diese Rezeptoren, um ein Lager an tumorreaktiven TCRs gegen Antigene zu füllen, die bei verschiedenen Patienten beobachtet wurden, und die in Zukunft als Standardtherapeutika eingesetzt werden könnten. Um personalisierte TCR-basierte Therapien zu ermöglichen, arbeiten wir daran, die Entdeckung von Rezeptoren mit patientenindividuelle Neoepitopen zu beschleunigen.
Personalisierte transgene T-Zell-Therapien können - in einigen Fällen - einen Patienten von Krebs heilen. Bis vor kurzem dauerte die Identifizierung patientenspezifischer, tumorreaktiver T-Zell-Rezeptoren jedoch über 3 Monate. Wir verkürzen diese Identifizierungsphase auf nur wenige Wochen, indem wir Einzelzellsequenzierung, maschinelles Lernen und halbautomatisches Klonen im Hochdurchsatzverfahren kombinieren. Wir arbeiten eng mit dem Team für T-Zell-Therapieentwicklung zusammen, um neue Therapien für Hirntumorpatienten zu entwickeln.
Unser Modell für maschinelles Lernen ist für die akademische Nutzung unter: https://predicTCR.com, verfügbar, für die kommerzielle Nutzung kontaktieren Sie unser Spin-off: https://www.tcelltech.eu/.
Wir sind in reversen translationalen Studien aktiv, entdecken tumorreaktive T-Zellrezeptoren in Proben aus den laufenden klinischen Studien des Labors und testen sie in präklinischen Modellen. Wir arbeiten daran, solche T-Zellrezeptoren zu handelsüblichen Zelltherapien für Hirntumorpatienten weiterzuentwickeln. In unserer bevorstehenden klinischen Studie INVENT4GB nutzen wir die neuesten Technologien, wie beispielsweise über episomale DNA-Vektoren vermittelte T-Zellrezeptoren. Darüber hinaus interessieren wir uns seit langem für die Rolle der HLA-Klasse II Moleküle in Hirntumoren, die Möglichkeit der Immunüberwachung von Liquorproben bei seltenen Krankheiten und das Verständnis des T-Zell-Traffics im Gehirn.
Unser Ziel ist es, zwei der Hauptmerkmale von immunologisch kalten Hirntumoren zu verstehen und zu bekämpfen, (i) den Ausschluss von T-Zellen aufgrund des Wachstums hinter der Blut-Hirn-Schranke in einem immunologisch speziellen Kompartiment und (ii) eine hochgradig immunsuppressive Mikroumgebung des zentralen Nervensystems, die zytotoxische T-Zellen hemmt, was beides zu einzigartigen Resistenzmechanismen führt. Die umfassende räumliche, transkriptomische und funktionelle Charakterisierung eines heterogenen und komplexen Zusammenspiels von T-, myeloischen und Stromazellen weist den Weg zu neuartigen kombinatorischen Therapiekonzepten, die in verschiedenen präklinischen Hirntumormodellen untersucht werden.
Unsere Forschungsteams
Mehr Informationen finden Sie auf unserer englisch sprachigen Homepage:
Cell Therapy | German Cancer Research Center
Mehr Informationen finden Sie auf unserer englisch sprachigen Homepage:
Immunogenomics | German Cancer Research Center
Mehr Informationen finden Sie auf unserer englisch sprachigen Homepage:
Immune Microenvironment | German Cancer Research Center
Mehr Informationen finden Sie auf unserer englisch sprachigen Homepage:
Immunotherapy Brain Tumor Models | German Cancer Research Center
Mehr Informationen finden Sie auf unserer englisch sprachigen Homepage:
Immune monitoring and TCR discovery | German Cancer Research Center
Ausgewählte Publikationen
Nature Communications
Cancer Cell
L. Bunse, T. Bunse, M. Kilian, F. Quintana, M. Platten
Neues aus dem Labor
Mitarbeiter
-

Prof. Dr. med. Michael Platten
Department Head
-

Belize Acharya
MD-Student
-

Dr. Dennis Alexander Agardy
PostDoc
-

Kuralay Aman
MD Student
-

Dr. Gabor Bakos
PostDoc
-

Dr. Theresa Bunse
Teamleader
-

Dr. Lukas Bunse
Teamleader
-
Luis Diel
-

Amelie Christina Dietsch
MD Student
-

Andreas Dobbelstein
MD Student
-

Greta Döking
Research Technician IMU
-

Fabian Edmeier
MD Student
-

Alexander Ernst
Technical Assistance IMU
-

Montserrat Escobar Rosales
Intern
-

Henrike Feldmann
PhD Student
-

Hannah Gelhaus
PhD Student
-

Mehsoon Gilani
PhD Student
-
Krish Gopalan
-

Niklas Graßl
PostDoc
-

Dr. Edward Green
Teamleader
-

Sofia Heras Valdivielso
Intern
-

Julian Hlawatsch
MD Student
-

Zeren Hu
MD Student
-

Lena Hönig
Secretary
-
Melissa Höpfner
Technical Assistance
-

Ingrid Hülsmeyer
Technical Assistance IMU
-
Gyuyeon Jang
Intern
-

Kristine Jähne
Technical Assistance
-

Simone Jünger
Technical Assistance IMU
-

Vahid Khaki Bakhtiarvand
PhD Student
-

Philipp Koopmann
MD Student
-

Dr. Tom Niklas Kuhn
PostDoc
-
Ella Kuipers
Intern
-

Dr. Katharina Lindner
PostDoc
-

Claudia Maldonado Torres
Technical Assistance
-

Dr. Iris Mildenberger
Teamleader
-

Sarwar Mustafa
MD Student
-

Marie-Therese Neuhoff
PhD Student
-

David Palmero Canton
PhD Student
-

Alina Paul
PhD Student
-

Dr. Isabel Poschke
Teamleader
-

Fatemeh Rezazadeh
Intern
-

Panthea Ruhdorfer
MD Student
-

Samira Moreen Sachse
Technical Assistance IMU
-

Dr. Katharina Sahm
Teamleader
-

Georgios Samaras
PhD Student
-

Khwab Sanghvi
PostDoc
-

Robin Seitz
Technical Assistance
-

Akanksha Shukla
Technical Assistance IMU
-

Aleksa Simic
Technical Assistance
-

Saskia Stange
PhD Student
-

Dr. Chin Leng Tan
PostDoc
-

Clara Tejido Dierssen
PhD Student
-
Ceyda Tozar
Student Assistant
-

Martin Uerlich
PhD Student
-

David Vonhören
PostDoc
-

Dr. Marie-Christine Wagner
Lab and project management
-

Binghao Zhao
MD Student
Kontaktieren Sie uns





































