Fighting cancer with viruses therapeutic applications of adenoviruses
Cancer is still a leading cause of death and treatment options are unsatisfactory for many malignancies. Advances in molecular and cellular oncology together with new and powerful genetic technologies now offer the opportunity for the rational development of new cancer therapies. Viruses have become of high interest for cancer treatment by two different therapeutic strategies: gene therapy and virotherapy. In gene therapy, engineered viruses are exploited as vectors for transfer of therapeutic genes into patients cells. An example is the transfer of genes encoding tumor-antigens for genetic vaccination. Virotherapy, or viral oncolysis, is the killing of tumor cells by virus infection using engineered viruses that feature tumor-selective replication. Thus, this approach implements a new mode of tumor cell killing (viral cell killing), facilitates amplification of the drug in the patients tumors (virus replication) and allows for the adaptation of the drug for specific tumor types or treatment regimens (engineering of the virus). Adenoviruses are non-enveloped viruses with a double-strand DNA genome and a lytic replication cycle. The exceptional knowledge of their structure, genome organization and replication cycle has facilitated their development as gene transfer vectors and oncolytic viruses.
Challenges for virotherapy with oncolytic adenoviruses
First generation oncolytic adenoviruses have been investigated in several clinical trials. These have shown that side effects are tolerable, even after systemic injections of adenoviruses at high titers. A further important result was the observation that at least under certain circumstances - oncolytic adenoviruses can destroy tumors in patients. However, overall therapeutic efficiency of first generation oncolytic adenoviruses was insufficient. Thus, the major challenge for ongoing research is to derive oncolytic adenoviruses with improved therapeutic potency.
Our current research topics:
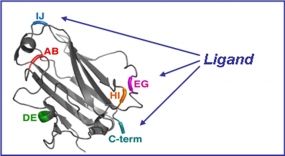
Structure of the cell-binding domain of the adenovirus capsid. Positions investigated for the insertion of tumor-specific peptide ligands are highlighted in color.
© Nettelbeck
Capsid Engineering
Adenoviruses feature a protein capsid, which is an icosahedron with 12 antennae-like penton proteins in the vertices. These pentons are responsible for cell binding and entry. We genetically engineer these proteins to redirect adenovirus cell binding and entry (tropism-modification). Our goal is to prevent viral entry into healthy cells and enhance infection of tumor cells.
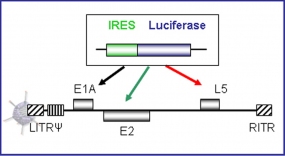
Strategy for transgene insertion into different early and late adenoviral transcription units exploiting an internal ribosome entry site (IRES).
© Nettelbeck
Transgene expression
To combine adenoviral oncolysis with gene therapy, we insert therapeutic genes into the virus genome. In this regard, we investigate (i) strategies for efficient and non-disturbing transgene expression by replication-competent adenoviruses and (ii) different candidate therapeutic genes. This project aims at the killing of tumor cells that are not killed by the virus, for example due to anatomical barriers which can prevent virus spread in the tumor.
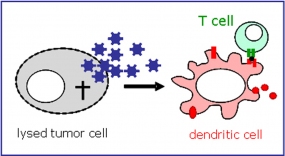
Model for anti-tumor immune activation by professional antigen-presenting cells (dendritic cells) after uptake of melanoma cell lysate.
© Nettelbeck
Anti-tumor immune activation
To date, it is not known whether adenoviral oncolysis induces immune responses to the tumor cells and, if so, whether such immune responses have potent therapeutic activity. We investigate how adenoviral tumor cell killing modulates immune cell functions and develop strategies to improve oncolytic adenoviruses for enhanced anti-tumor immune activation. The goal is the implementation of a viro-/immunotherapy to combine potent tumor cell killing by adenoviral oncolysis with systemic and prolonged anti-tumor immunity.

