Research for a life without cancer
At the DKFZ, we want to ensure that fewer people develop cancer, that cancer can be cured or treated so effectively that those affected can live with the disease and grow old with a good quality of life.
About DKFZ







Latest from DKFZ
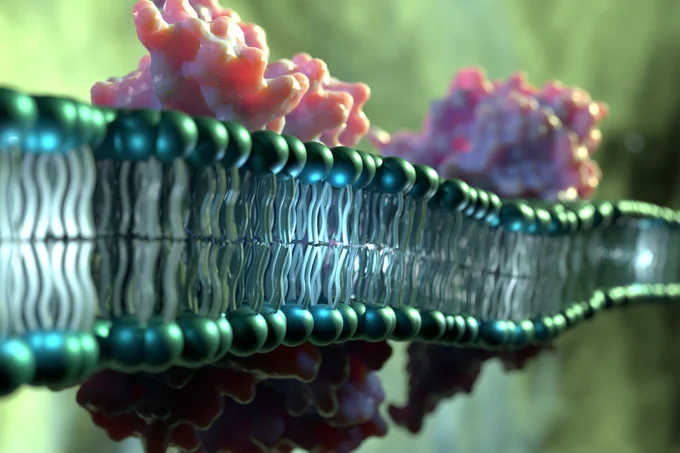
In response to stress or damage, cells undergo senescence and stop dividing. However, if senescent cells accumulate in tissues over the long term, chronic inflammation occurs and the risk of cancer increases. Researchers at the German Cancer Research Center (DKFZ) have now discovered a previously unknown mechanism by which senescent cells protect themselves from oxidative stress and a specific form of cell death known as ferroptosis. In the long term, these findings could provide new avenues for cancer therapies and the treatment of age-related diseases.




Our research opens doors in the fight against cancer


Do you have questions on the topic of cancer?
Let us advise you!
Doctors from the Cancer Information Service answer your questions every day. Find out more now for free!