Slimming Aid from the Cell Laboratory? Inflammation enzyme regulates the production of brown fat tissue
Scientists of the German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) have published an article in Science revealing that the COX-2 inflammation enzyme stimulates the formation of new brown fat tissue in mice. Brown fat tissue transforms energy into heat. Therefore, mice with increased COX-2 production have a higher energy consumption and are slimmer. On the basis of these results scientists might develop a novel weight loss method for pathogenic obesity.
Love handles, muffin tops and stomach tires white fat tissue forms the typical curves in the notorious problem areas to store energy. Exactly the opposite happens in brown fat tissue: Instead of being stored, energy gets transformed into heat. To the dismay of many people, adults have only small amounts of this energy burner. By contrast, babies and animals in hibernation have lots of it in their bodies where it serves for heat regulation.
Researchers know that external influences can stimulate the production of brown fat tissue in animals. If rodents are kept at low temperatures, clusters of brown fat cells form amid the white fat tissue. A DKFZ research team headed by Dr. Stephan Herzig, jointly with colleagues from Munich, Marburg, Frankfurt and Lausanne, has investigated the molecular causes of this phenomenon. They discovered that the production of the COX-2 inflammation enzyme is increased in white fat tissue of mice after exposure to cold temperatures. COX-2 is well known to scientists: It regulates the key step in the biosynthesis of prostaglandins inflammation-promoting hormones which are also responsible for activating pain.
Our recent results prove that COX-2 and prostaglandins are crucial for the formation of new brown fat tissue and, thus, also for regulating body weight, said Stephan Herzig summarizing his data. Parallel to the increase in COX-2 production in white fat tissue, there is also a rising level of the protein which biochemically transforms energy into heat and is therefore considered the most important biomarker for brown fat cells. When the investigators switched off COX-2 in the white fat tissue, however, the typical appearance of brown fat cells could no longer be stimulated by the cold.
Even without using cold temperatures the scientists were able to stimulate the formation of brown fat cell clusters in white fat tissue by boosting the COX-2 production in mice using a molecular-biological trick. The body weight of these animals was 20 percent lower than that of normal animals. Even on a calorie-rich diet they did not put on weight.
Previous studies by researchers from Finland suggest that in humans, too, the activity, i.e. heat production of brown fat tissue can be stimulated by regular stays in the cold. Stephan Herzig now plans to investigate whether COX-2 also plays the biochemical key role here. Since the formation of brown fat cells can also be stimulated in the culture dish, Herzig sees a possibility for practical use of his results. His vision is to take white fat tissue from severely obese individuals, to stimulate the formation of brown fat cells using prostaglandins in the culture dish, and then transplant them back to the patient. Thus it would be possible to help people by boosting their energy metabolism and thus making weight loss easier. Scientists estimate that a small amount of about 50 grams of brown fat tissue would be sufficient to increase a persons energy metabolism by 20 percent. This would be equal to a reduction of body weight by 20 kilograms over a period of one year.
Clinical trials with cancer patients have already indirectly revealed that COX-2 has an influence on body weight. Thus, the body weight of patients suffering from severe cancer-related wasting stabilized due to COX-2 inhibiting drugs. But a number of common pain relievers also act by inhibiting COX-2. Does that mean that users have to accept weight gain as a side effect? Herzig has good news: So far, we have found no evidence suggesting that one has to fear gaining weight from taking these drugs.
A picture is available at the Internet:
http://www.dkfz.de/de/presse/pressemitteilungen/2010/images/Fettzellen.jpg
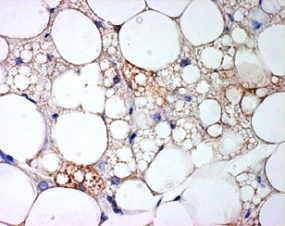
Picture caption: brown fat cells within white fat tissue
Picture Source: Karin Müller-Decker, Deutsches Krebsforschungszentrum
Alexandros Vegiopoulos, Karin Müller-Decker, Daniela Strzoda, Iris Schmitt, Evgeny Chichelnitsky, Anke Ostertag, Mauricio Berriel Diaz, Jan Rozman, Martin Hrabe de Angelis, Rolf M. Nüsing, Carola W. Meyer, Walter Wahli, Martin Klingenspor und Stephan Herzig: Cyclooxygenase-2 Controls Energy Homeostasis in Mice by de Novo Recruitment of Brown Adipocytes
Science 2010, DOI: 10.1126/science.1186034
With more than 3,000 employees, the German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) is Germanys largest biomedical research institute. DKFZ scientists identify cancer risk factors, investigate how cancer progresses and develop new cancer prevention strategies. They are also developing new methods to diagnose tumors more precisely and treat cancer patients more successfully. The DKFZ's Cancer Information Service (KID) provides patients, interested citizens and experts with individual answers to questions relating to cancer.
To transfer promising approaches from cancer research to the clinic and thus improve the prognosis of cancer patients, the DKFZ cooperates with excellent research institutions and university hospitals throughout Germany:
The DKFZ is 90 percent financed by the Federal Ministry of Education and Research and 10 percent by the state of Baden-Württemberg. The DKFZ is a member of the Helmholtz Association of German Research Centers.