Division of Medical Physics in Radiology
Prof. Dr. sc. techn. Mark E. Ladd
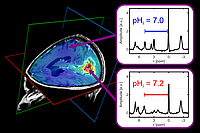
3D mapping of intracellular pH in a glioma patient using phosphorus-31 MR spectroscopic imaging at 7 Tesla (method according to Korzowski A et al., 2020, Magn Reson Med, 84:1707-1723). (Left) Interpolated pH maps overlaid on a clinical contrast-enhanced MR image. (Right) pH values are calculated via the chemical shift difference between phosphocreatine and inorganic phosphate.
© dkfz.de
The Division of Medical Physics in Radiology develops new methods for imaging-based diagnostic and therapeutic procedures. Our research focuses on novel hardware as well as software-based acquisition and reconstruction strategies for magnetic resonance imaging (MRI), positron emission tomography (PET), and optical tomography. We strive to improve and individualize cancer patient treatment by acquiring quantitative biomedical information about tumors and metastases with non-invasive imaging methods. For example, we are expanding the diagnostic value of MRI by using very powerful magnetic fields (7 or 9.4 Tesla) to depict the distribution of sodium, oxygen, potassium, and chlorine inside the body. Another approach to capture metabolic processes is hyperpolarization of carbon in various chemical substrates, which are then injected into the body and measured with MRI. By optimizing MRI diffusion techniques, we have been able to greatly improve the diagnostic accuracy of breast cancer screening, and we are investigating how maps of tissue susceptibility correlate with disease. An additional emerging MR imaging contrast is provided by Chemical Exchange Saturation Transfer (CEST) imaging, which allows detection and measurement of glucose or mobile proteins. Furthermore, we are exploring new detector concepts for simultaneous PET and optical imaging.
Medical imaging continues to be one of the key technologies for cancer detection, characterization, and therapy monitoring. Despite the enormous technological advances achieved in the past decades, imaging still has enormous potential to reveal more information about the metabolic, physiologic, and functional parameters of tumors and metastases. This information can then be used to choose the best therapy for each individual patient. In collaboration with clinical divisions of the DKFZ and partners at the university hospital, we are working to translate our developments into standard patient use. This includes state-of-the-art imaging protocols at our MR imagers located at the National Center for Tumor Diseases (NCT).

